Visceral Abdominal Pain
Author
Leonardo Kapural, MD, PhD
Professor
Department of Anesthesiology
Cleveland Clinic Lerner College of Medicine
Case Western University
Clinical Research Director
Department of Pain Management
Cleveland Clinic
Cleveland, OH
Introduction
Abdominal pain is one of the most frequent complaints to a primary care physician, accounting for nearly 2.5 million office visits a year.[1] In many patients who present with abdominal pain, a definitive diagnosis can be made, but in 35-51% of patients, no identifiable cause is found.[2] There are approximately 2 million patients in the United States who currently experience severe abdominal pain. Most of them undergo a multitude of imaging studies, consults to gastroenterology and surgery before being referred to a chronic pain specialist.[3-4]
Pathophysiology
Abdominal pain may not have an origin inside the abdomen, but rather within the abdominal wall and may be caused by localized muscle spasm or a nerve injury. The majority of currently diagnosed abdominal pains are of the visceral cause (intra-abdominal adhesions or pre-existing disease including inflammatory diseases such as Crohn’s disease and ulcerative colitis, diffuse endometriosis, etc.).
Chronic pancreatitis and various motility disorders like gastroparesis or irritable bowel syndrome are frequent causes of abdominal pain in any pain medicine clinic. Somatosensory abdominal wall pain can be sometimes confused with the visceral pain origin and is frequently caused by the neuralgias of the illiohypogastric, illioinguinal or genitofemoral nerve as well as some clinically less obvious intercostal neuralgias. In addition, it can be myofascial and inflammatory like an epigastric pain in chronic costochondritis (Tietze’s disease).
To make diagnosis even more difficult, long-standing abdominal, visceral pain and repetitive stimulation may produce the changes in the size of cutaneous receptive fields of the abdomen. The phenomenon of viscero-visceral convergence with so-called referred visceral hyperalgesia/allodynia occurs when one intraabdominal visceral pain source potentiates the pain from another visceral source. Viscero-somatic convergence means that one visceral source of pain may produce referred cutaneous (skin) hyperalgesia/allodynia and/or referred muscle hyperalgesia/allodynia.
The above listed phenomena illustrate the complexity of visceral referral patterns and resultant difficulties in establishing diagnosis when the source of abdominal pain is unclear.
Clinical Features
When the patient presents to a pain management office, a thorough history is obtained and a physical exam is performed to determine the source and/or nature of the pain.
Physical examination may help to determine the source of the abdominal pain. It can also provide supportive evidence for the diagnosis by excluding other conditions. Some of the serious signs such as significant weight loss, an abdominal mass, or blood in the stool should prompt further investigation. Inspection may show surgical scars indicating previous laparotomies. Allodynia and/or hyperalgesia along the scar may suggest presence of nerve damage and/or neuroma. Abdominal wall pain must be excluded by palpation and performance of the Carnett’s test (pain from palpation increases with raising the head and contracting the rectus abdominis muscle). However, keep in mind that viscerosomatic referral from some of the functional motility disorders could mimic abdominal wall pain.
Listening and responding to the patient’s concerns and expectations is very important, especially if the cause of abdominal pain is unclear. Additional tests may be ordered on the basis of objective data rather than pain severity. Psychosocial assessment, preferably including objective tests like depression inventories, is an important tool which may direct future treatment.
Diagnostic Examination
Patients frequently undergo an extensive work-up to determine the source of their abdominal pain. Still, in a significant group of the patients, a search for the clear cause of their visceral pain remains elusive. An algorithm on a decision-making process for diagnostic examination in patients with chronic abdominal pain is shown in Figure 1.
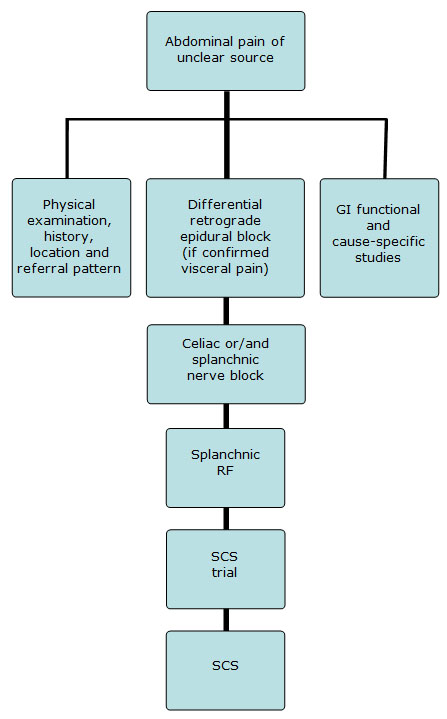
Figure 1. Proposed decision-making and interventional pain management algorithm for the treatment of severe abdominal pain non-responsive to previous conservative therapies.
Diagnostic Epidural Block
A differential retrograde epidural block can be useful to help determine the source of the abdominal pain.[5] This test is similar to the diagnostic spinal test that Winnie recommended in the 1970s. During the diagnostic differential epidural block, an epidural catheter is placed, preferably under fluoroscopy. The patient is then brought to the recovery room or other monitored area where the test will be conducted. The physician injects various medications (local anesthetics, saline etc) via the catheter at different intervals during this diagnostic test. Local anesthetic (usually 2% 3-chlorprocaine or 1% lidocaine 10-30 mL) serves to differentiate between predominantly visceral, somatosensory or central chronic pain, while placebo injection may help to differentiate placebo responders, malingering and sometimes psychogenic source of patient’s chronic pain.
The sensitivity and specificity of such retrograde differential epidural blocks is relatively low. Still such test, together with the other diagnostic findings like imaging, diagnostic blocks and physical exam may contribute to the establishment of a diagnosis. Further diagnostic blocks used to distinguish actual source of the truncal pain may include diagnostic intercostals nerve blocks or trigger point injections if differential nerve block suggested somatosensory source of the patient’s pain or splanchnic, celiac or hypogastric if visceral source is suspected.
Treatment
Celiac Plexus Block
If the results of the differential block reveal that the pain is of the visceral origin then the physician may proceed with a celiac plexus block or a splanchnic nerve block. A celiac plexus block is performed most commonly for the treatment of upper and mid-abdominal pain and is most effective in relieving the pain from malignancies of the pancreas, liver, gallbladder, omentum, mesentery, and alimentary tract from the stomach to the large colon. It has also been used to control chronic non-malignant pain from various causes including chronic pancreatitis and different motility causes of abdominal pain. The procedure has been found to lessen or eliminate upper abdominal pain and it can be used by the physician as a diagnostic nerve block to further differentiate visceral from somatic pain.
Classical description of celiac plexus block involves placement of the needle through the paraspinal area of the middle back (L1 vertebral body). Fluoroscopy is used to facilitate placement of the 22-24 G needle to the anterolateral area of the first lumbar vertebral body. Fifteen to 20 mL of 0.375% bupivacaine or some other local anesthetic is delivered bilaterally with prior instillation of the contrast to confirm appropriate placement of the drug (Figure 2). The procedure is low risk, non-surgical, and can be an effective alternative when conservative treatments have been exhausted or side-effects from the oral medications cannot be tolerated by the patient. The most common side effects from a celiac plexus block include local pain, hypotension and diarrhea.[6]
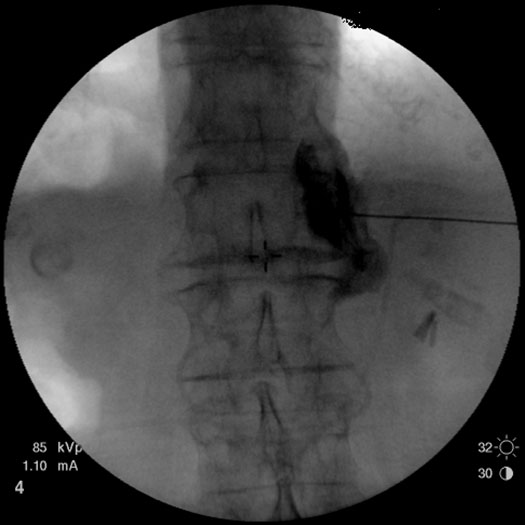
Figure 2. A right celiac plexus with the contrast medium showing mostly a right-sided spread.
Splanchnic Nerve Block
Two 22-24 G needles are positioned on each side of the spine (Figure 3A) to target the sympathetic nerves (greater and lesser splanchnic nerve). Once the needle is in position, contrast is delivered (Figure 3B) and spread confirmed within the desired area. Immediately after, local anesthetic is injected to block the nerve fibers.
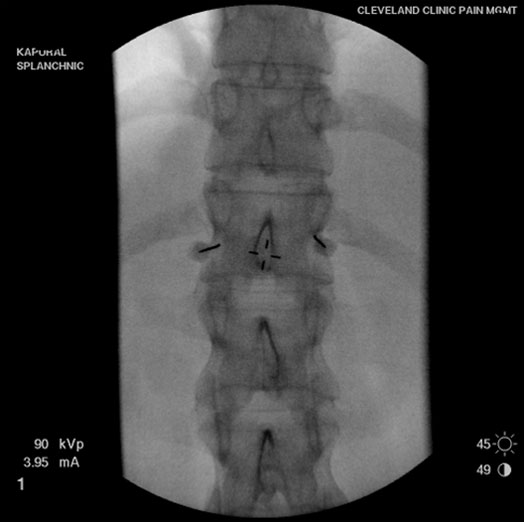
Figure 3A. Needles placed on each side for bilateral splanchnic nerve blocks.
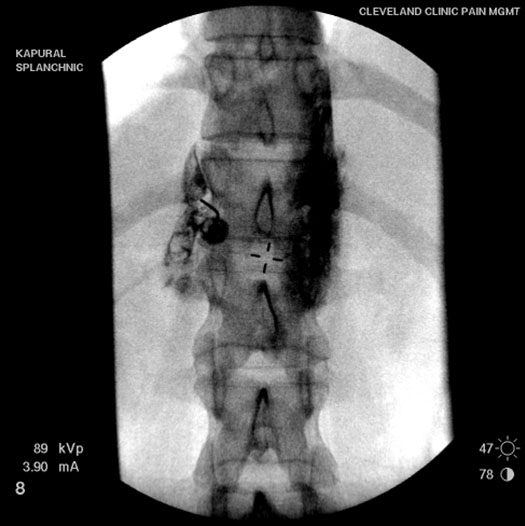
Figure 3B. Injection of contrast medium showed spread along the anterolateral aspect of the vertebra.
The splanchnic nerve block has been found to provide pain relief in patients who fail to obtain relief or only achieve short term (7 The splanchnic nerves transmit the majority of the nociceptive information from the viscera. The splanchnic nerve block specifically interrupts sympathetic nerve transmission to the celiac plexus without interrupting the abdominal parasympathetic nerves.
The technique for splanchnic nerve block differs little from a celiac plexus block except that the needles are aimed more towards the head or anterior section of the nerve plexus.[8] The final position of the needle tip is just above the diaphragm which allows the physician to block the greater, lesser, and least splanchnic nerves before they cross the diaphragm into the abdomen. The risks of a splanchnic nerve block are similar to those of a celiac plexus block. It is advisable to perform both procedures under fluoroscopic or CT guidance as accurate needle placement is needed to decrease the risk of pneumothorax, thoracic duct injury and inadvertent spread of the injected medications.[7]
Radiofrequency Ablation of Splanchnic Nerves
Depending on the results of a splanchnic nerve block a physician may decide to do a splanchnic radiofrequency ablation (RFA).5-7 The positioning of the needle for RFA is similar to the splanchnic nerve block, and should be performed under fluoroscopy guidance (Figure 4). The radiofrequency probe is heated to 80ºC over 90 seconds; a second lesion can be performed with the tip of the needle tip in a different position.
A radiofrequency electrode is inserted through the needle and a stimulation process is performed to help determine optimal placement of the electrode for the radiofrequency. Once correct placement is verified, a small radiofrequency current is sent through the electrode into the surrounding tissues, causing the tissue to heat. Mild discomfort in the back may be experienced following the procedure. Leg weakness or numbness, if it occurs, should last only for a few hours; the patient should only walk with assistance during that time. Discomfort in the back may occur when the local anesthetic wears off. This usually lasts two or three days and if it occurs, ice should be applied to the area the day of the procedure and moist heat the day after the procedure if the discomfort persists.[7]
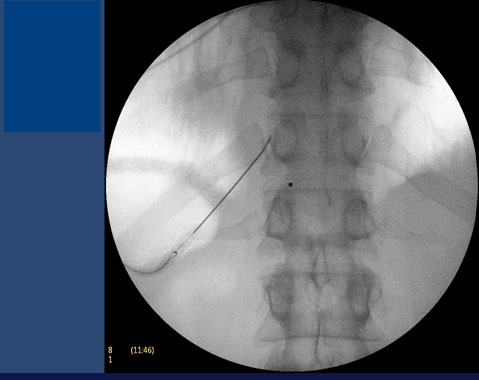
Figure 4. Radiofrequency ablation of splanchnic nerves.
Spinal Cord Stimulation
If temporary or inadequate pain relief is achieved following a celiac plexus, splanchnic nerve block, or splanchnic radiofrequency ablation, then spinal cord stimulation can be tried as another treatment option for the intractable abdominal pain. There is only a limited experience with spinal cord stimulation for the treatment of chronic visceral pain. Recent basic-science studies suggest that dorsal-column electromodulation may have a role in the treatment of visceral abdominal and pelvic-repeated distension and hyperalgesia.[8] In fact, there are several potential mechanisms by which spinal cord stimulation might suppress visceral pain.[8]
There are three steps to having implantation of a spinal cord stimulator (Figure 5). The first step is to a trial period to determine if spinal cord stimulation is effective. This trial period lasts about one to two weeks; the patient is not permitted to shower or take a bath as the bandages must remain dry. Rather, they are allowed to sponge bath. To avoid the risk of infection, an antibiotic is given for 7-10 days. The stimulator is removed at the end of the trial; an effective trial is a reduction of the patient’s pain by at least 50%.[9-13]
If the trial was successful then the next step is implantation of the device. The patient is asleep under deep sedation and a small incision is made in the buttock for placement of the battery and another incision is made in the back for implantation of the lead. While electrical stimulation prevents pain signals from the abdomen, it does not conceal pain from acute abdominal conditions (e.g. infection, inflammation or ileus).
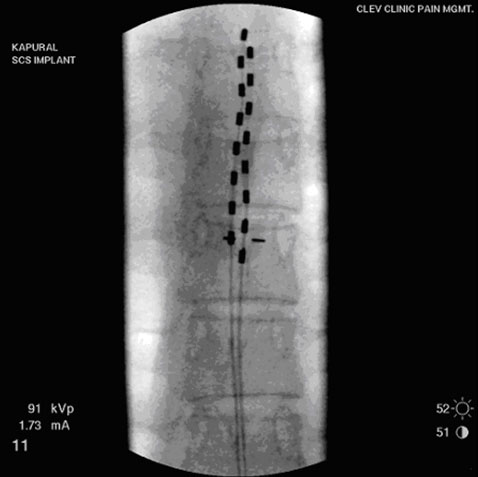
Figure 5. Electrical leads placed within epidural space in the spine.
Previously published small case series studies of SCS for abdominal and pelvic visceral pain have shown encouraging improvements in pain scores,[9-14] improved functional capacity,[9] and reduced opioid use.[9-13] Given the dismal history of conventional treatment for chronic visceral pain, these data suggest that spinal cord stimulation may be a useful alternative.
Summary
Comprehensive pain management is indicated for patients experiencing chronic visceral pain that interferes with activities of daily living. Multiple interventional options are available to improve symptoms of chronic visceral hyperalgesia and to possibly decrease opioid requirements. Visceral blocks and radiofrequency procedures can provide an extended period of pain relief. Spinal cord stimulation has emerged as a great therapeutic option for severe chronic abdominal pain. Intrathecal infusion is rarely needed.
References
- Everhart J. Overview. In: Everhart J, ed. Digestive diseases in the United States: epidemiology and impact. Vol. NIH publication no. 94-1447. Washington, D.C.: US Department of Health and Human Services, Public Health Service, National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases. 1994:3-53
- Klinkman MS. Episodes of care for abdominal pain in a primary care practice. Arch Fam Med 1996;5:279-85
- Russo MW, Wei JT, Thiny MT, Gangarosa LM, Brown A, Ringel Y, Shaheen NJ, Sandler RS. Digestive and liver diseases statistics. Gastroenterol 2004;126:1448-1453.
- Derbyshire SW. Imaging viseral pain. Curr Pain Headache Rep 2007;11:178-182
- Conwell DL, Vargo JJ, Zuccaro G, Dews TE, Mekhail N, Scheman J, Walsh RM, Grundfest-Broniatowski SF, Dumot JA, Shay SS. Role of differential neuroaxial blockade in the evaluation and management of pain in chronic pancreatitis. Am J Gastroenterol 2001;96:431– 436
- Erdine S. Celiac ganglion block. Agri 2005;17:14-22
- Raj PP, Sahinler B, Lowe M. Radiofrequency lesioning of splanchnic nerves. Pain Pract. 2002;2:241-247
- Krames ES, Foreman R. Spinal cord stimulation modulates visceral nociception and hyperalgesia via the spinothalamic tracts and the postsynaptic dorsal column pathways: A literature review and hypothesis. Neuromodulation 2007;10:224-237
- Kapural L, Janicky T, Narouze S, Mekhail N. Spinal cord stimulation is an effective treatment for the severe pelvic visceral pain. Pain Med 2006;7:440-3
- Krames E, Mousad DG. Spinal cord stimulation reverses pain and diarrheal episodes of irritable bowel syndrome: a case report. Neuromodulation 2005,8:82-88.
- Khan YN, Raza SS, Khan EA. Application of spinal cord stimulation for the treatment of abdominal visceral pain syndromes: Case reports Neuromodulation 2005;8:14-2
- Tiede JM, Ghazi SM, Lamer TJ, Obray JB. The use of spinal cord stimulation in refractory abdominal visceral pain: case reports and literature review. Pain Pract 2006 6:197-202
- Kapural L, Rakic M. Spinal cord stimulation for chronic visceral pain secondary to chronic non-alcoholic pancreatitis: a case report. Clin Gastroenterol Hepato 2008;42:750-751
- Kapural L. Spinal cord stimulation for Gastrointestinal painful disorders. In Textbook of Neuromodulation Eds. Krames E, Peckham H and Rezai A. 2009; pp 873-879
Leave a commentOrder by
Newest on top Oldest on top