How I Do It: Neuraxial Ultrasound
Cite as: Kolli S, Narouze S, Kalagara H. How I do it: neuraxial ultrasound. ASRA News 2021;46. https://doi.org/10.52211/asra110121.068
Central neuraxial blockade (CNB) with epidural or spinal anesthesia or analgesia is an established technique for surgical anesthesia and postoperative pain management. Traditional landmark-based palpation in tandem with loss of resistance to saline or air is highly successful for epidural placement in patients with normal anatomy.1 Similarly, spinal anesthesia can be performed using landmarks with needle tactile response and flow of cerebrospinal fluid serving as a clear endpoint. However, factors like obesity, congenital or acquired spine deformity, and previous spine surgery can pose technical challenges, but ultrasound can overcome inaccuracies in estimation of intervertebral space by palpation methods.2
Ultrasound imaging is used both before and during CNB procedures.3 Preprocedural scanning prior to skin puncture helps identify midline, appropriate interspace, any abnormal anatomy, depth to the epidural space, and planned needle trajectory.4,5 Then, real-time ultrasound-guidance is used to perform CNB.6 This article describes the anatomy of the spine, relevant ultrasonographic views, and the techniques used to perform neuraxial blocks using ultrasound imaging.
Gross Anatomy and Sonoanatomy of the Lumbar Vertebrae
Comprehensive knowledge of gross and sonoanatomy of the vertebrae and the vertebral canal is necessary to understand neuraxial imaging. Each lumbar vertebra is made up of a body and arch. The arch is comprised of pedicles, a spinous process, transverse processes, lamina, and superior and inferior articular processes (Figure 1). The vertebral canal or spinal canal is formed by the spinous process and lamina posteriorly, pedicles laterally, and vertebral bodies anteriorly. The thecal sac and its contents (ie, spinal cord, cauda equina, and cerebrospinal fluid) lie within the vertebral canal. The epidural space lies outside the thecal sac within the vertebral canal. Recognizing these key anatomical structures in parasagittal and transverse views facilitates performance of ultrasound-guided neuraxial interventions.
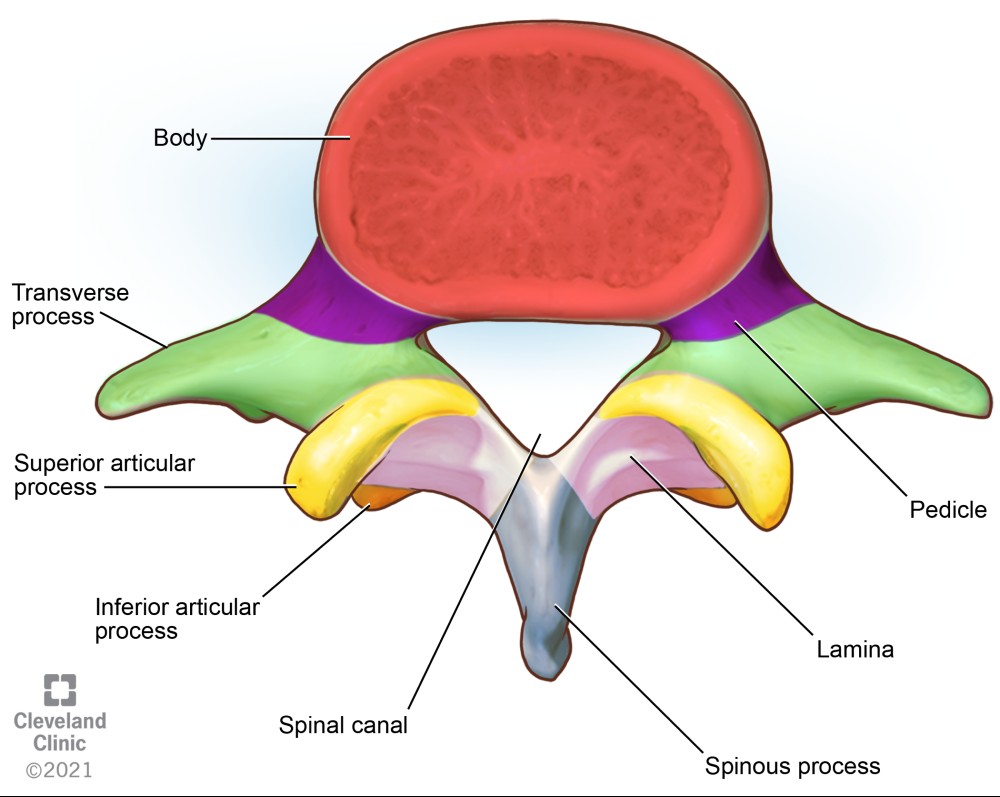
Figure 1. Superior view of a lumbar vertebra
Ultrasound cannot penetrate the bony surfaces of the lumbar vertebrae; hence they appear as hyperechoic white lines, casting dense acoustic shadows that have characteristic patterns corresponding to the bony contours. Interlaminar space, located posteriorly between the two adjacent vertebral laminae, is a bony window through which the ultrasound beam can pass through and enable the thecal sac to be visualized (Figure 2). The intervertebral foramina are located laterally between the two adjacent vertebrae where the spinal nerve roots emerge (Figure 3).
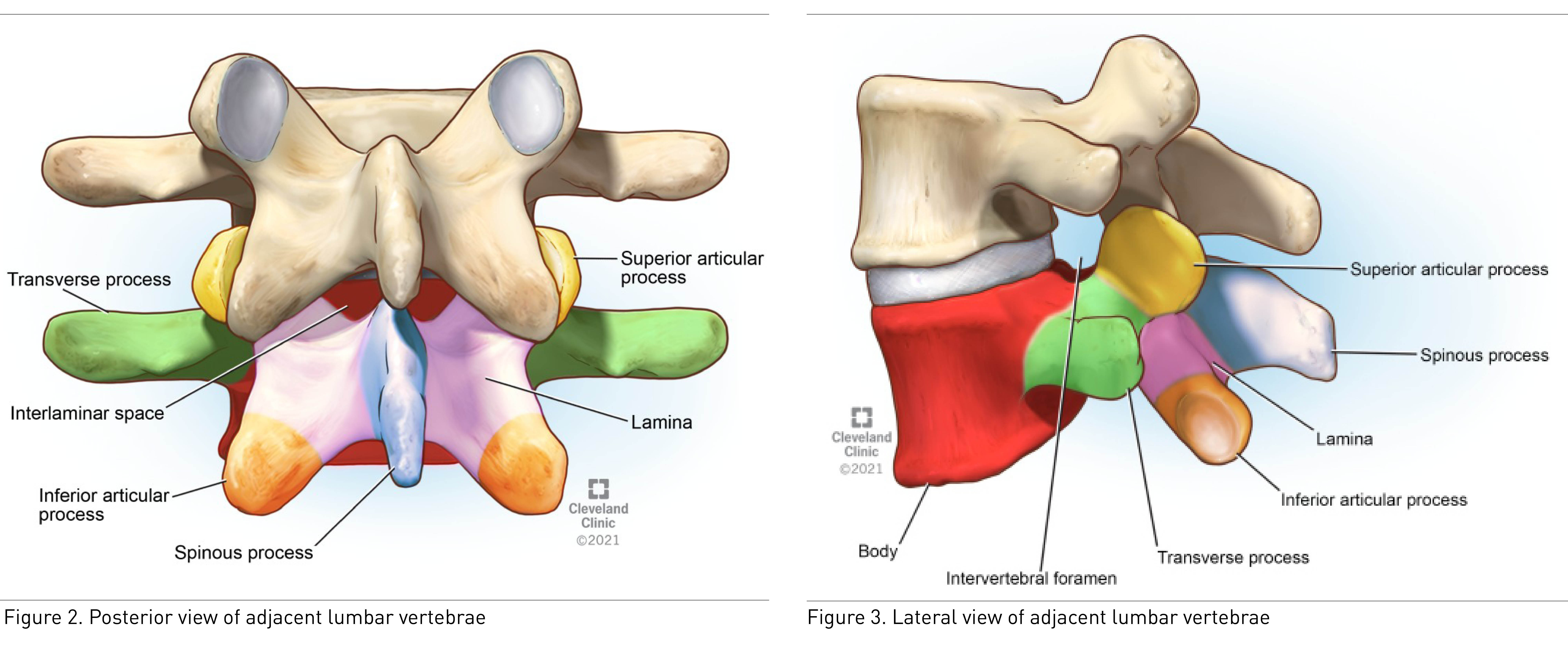
The ligamentum flavum, epidural space, and posterior dura often appear as single or sometimes double hyperechoic white structure, referred to as the posterior complex (PC). The anterior dura, posterior longitudinal ligament, and posterior aspect of the vertebral body are visible as a single hyperechoic white line, referred to as the anterior complex (AC).
Sonographic Views of the Lumbar Vertebrae
We use a curved-array, low-frequency (2–5 MHz) ultrasound transducer for neuraxial scanning with the patient in the sitting or lateral decubitus position. A low-frequency transducer allows for deeper penetration and wider field of view, improving the image quality and anatomy recognition. We conduct preprocedural scanning routinely for all patients prior to epidural and spinal anesthesia. This allows clinicians to become comfortable with spine imaging and pattern recognition in patients with normal anatomy and may improve success in challenging scenarios.
Accurate estimate of intervertebral level, location of midline, measurement of the depth to the epidural space and identification of other relevant structures requires systematic scanning in sagittal-oblique (Figure 4) and transverse (Figure 5) planes. Seven ultrasonographic views of the lumbar spine are described for comprehensive neuraxial scanning7 (Figure 6). The midline sagittal spinous process view can be used to identify the midline, whereas transverse oblique foraminal view is used for chronic pain procedures. Conventionally, three parasagittal and two transverse views are performed for neuraxial blockade and are described below.
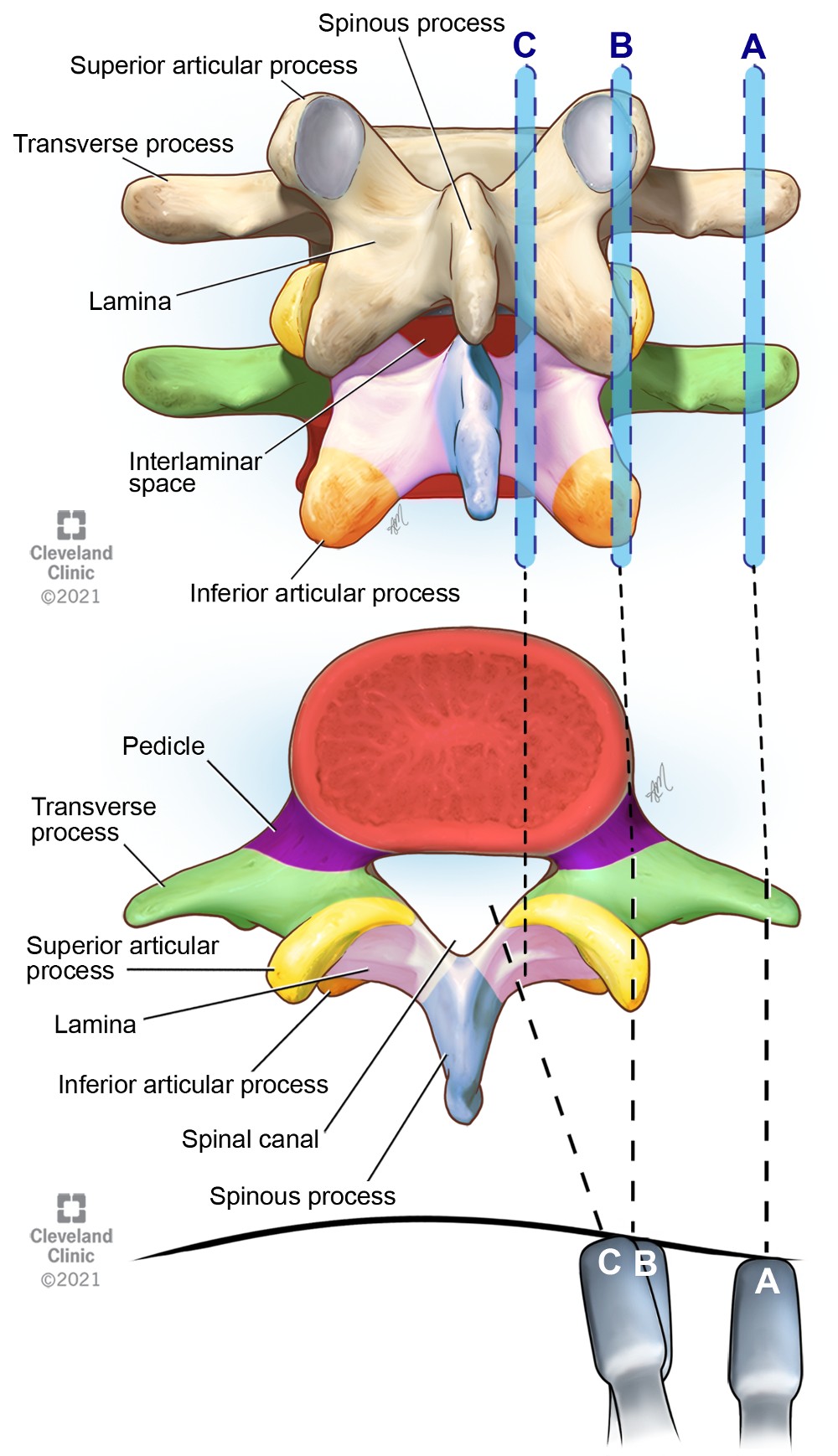
Figure 4. Parasagittal views of the lumbar vertebrae showing transducer positions A, B, and C
(A) Parasagittal transverse process view of the spine showing transverse processes
(B) Parasagittal articular process view showing the articular processes
(C) Parasagittal oblique interlaminar view showing the interlaminar acoustic windows
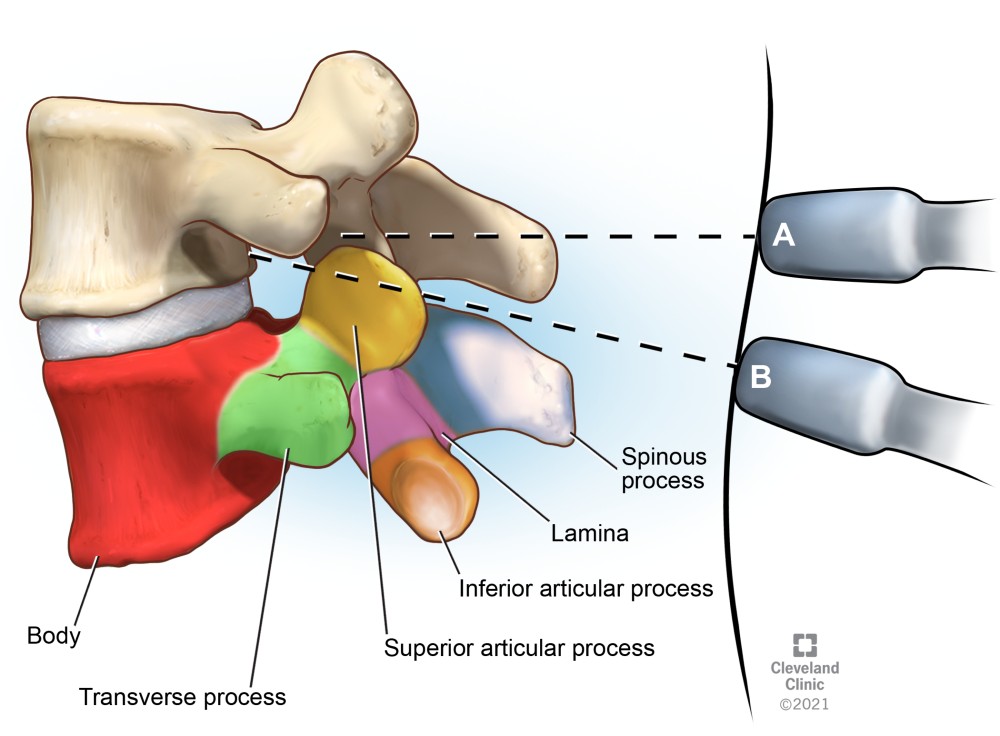
Figure 5. Transverse views of the lumbar vertebrae transducer positions
(A) Transverse spinous process view
(B) Transverse interlaminar (interspinous) view
- Parasagittal transverse process view
- Parasagittal articular process view
- Parasagittal oblique interlaminar view
- Sagittal spinous process view
- Transverse spinous process view
- Transverse interlaminar (interspinous) view
- Transverse oblique foraminal view
Figure 6. Ultrasonographic Views of the Lumbar Spine
Parasagittal Transverse Process View
Place the ultrasound transducer in a parasagittal plane, a few centimeters lateral to the midline and cephalad to the sacrum (Figure 4, probe position A; Figure 7A). The transverse processes of successive vertebrae appear as hyperechoic curvilinear structures with deeper hypoechoic shadows as dark finger-like projections, often described as a “trident sign” (Figure 7B). The psoas major muscle is seen deep and between the hypoechoic shadows, whereas the erector spinae muscle group is superficial. The view can be used to estimate the accurate vertebral level before the neuraxial procedure.
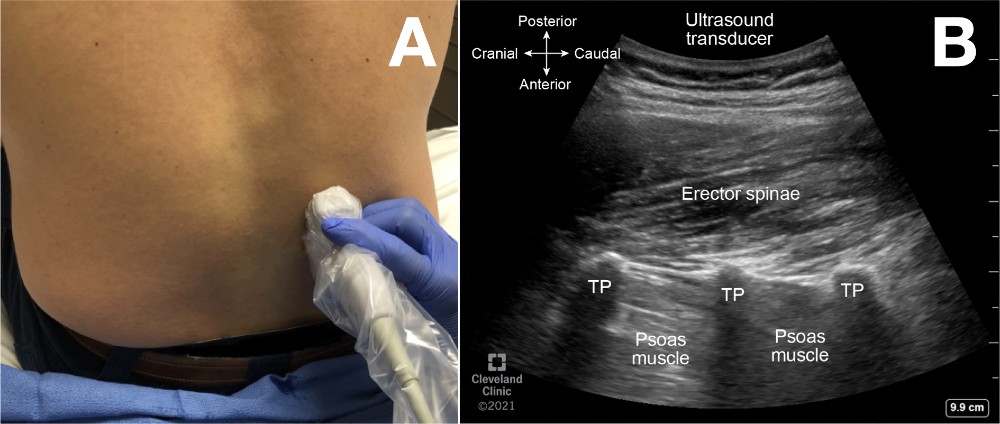
Figure 7. Parasagittal transverse process view
(A) Probe position to obtain parasagittal transverse process view of lumbar spine
(B) Parasagittal transverse process view (trident sign) ultrasound image
The exact vertebral level can be identified using the parasagittal scanning technique. Place the ultrasound transducer over the sacrum to identify the flat sacral bone, L5 transverse process and L5–S1 intervertebral space (Figure 8). Slide the transducer cranially to identify the respective L5–L4, L4–L3, and L3–L2 interspaces, and mark the desired space(s).
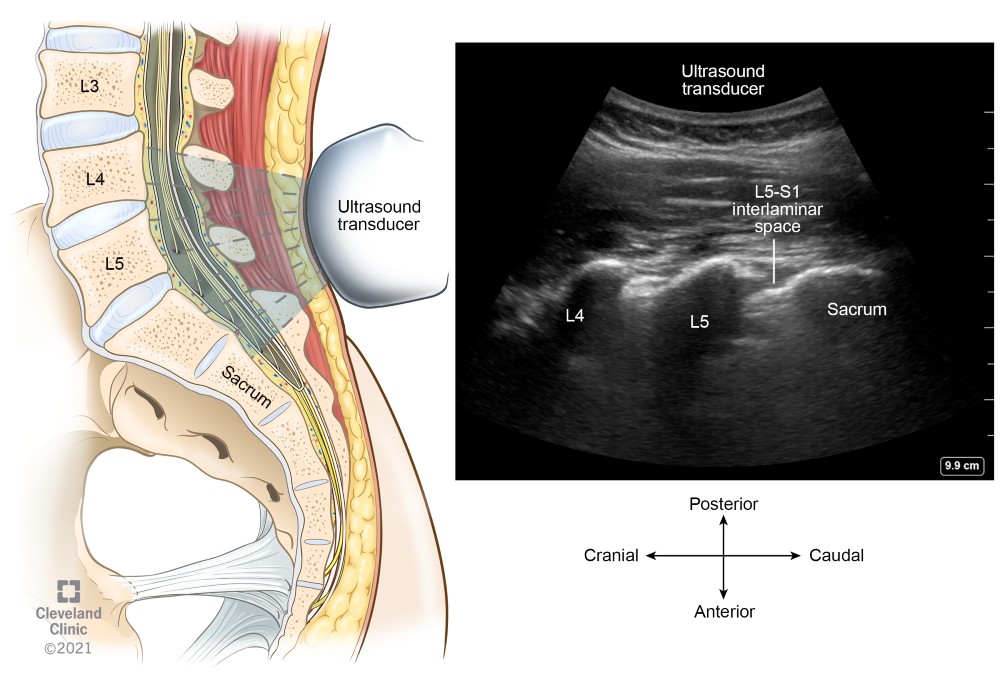
Figure 8. Parasagittal view of the sacrum and lower lumbar vertebra showing L5–S1 interlaminar space
Parasagittal Articular Process View
From the parasagittal transverse process view, slide the transducer medially until a continuous white hyperechoic line of humps is evident (Figure 9). Humps are the acoustic shadows of the articular processes of the facet joints, described as “camel hump sign” (Figure 4, probe position B). Neuraxial structures are difficult to see in this view because the bony structures are continuous and does not permit ultrasound signals beyond the articular processes.
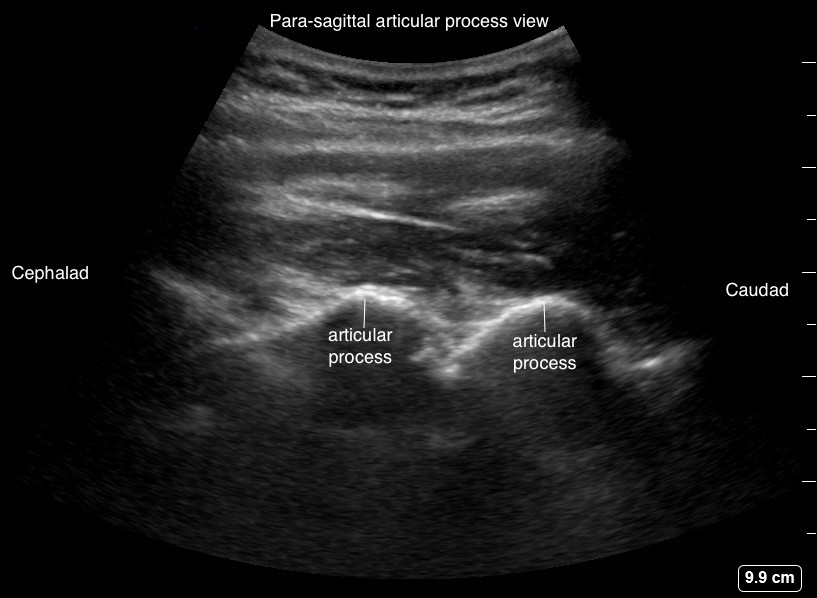
Figure 9. Parasagittal articular process view (camel hump sign)
Parasagittal Oblique Interlaminar View
From the parasagittal articular process view, tilt the transducer medially toward the median sagittal plane and bring the lamina into view (Figure 4, probe position C). The sloping hyperechoic laminae of adjacent lumbar vertebrae with gaps in between form a sawtooth or horsehead pattern (Figure 10). The gaps represent the interlaminar spaces through which the PC and AC may be visualized. This is the most important view in sagittal scanning to identify interspaces and mark the skin appropriately. This view is also helpful in identifying the most patent spaces for a paramedian approach to a neuraxial procedure.
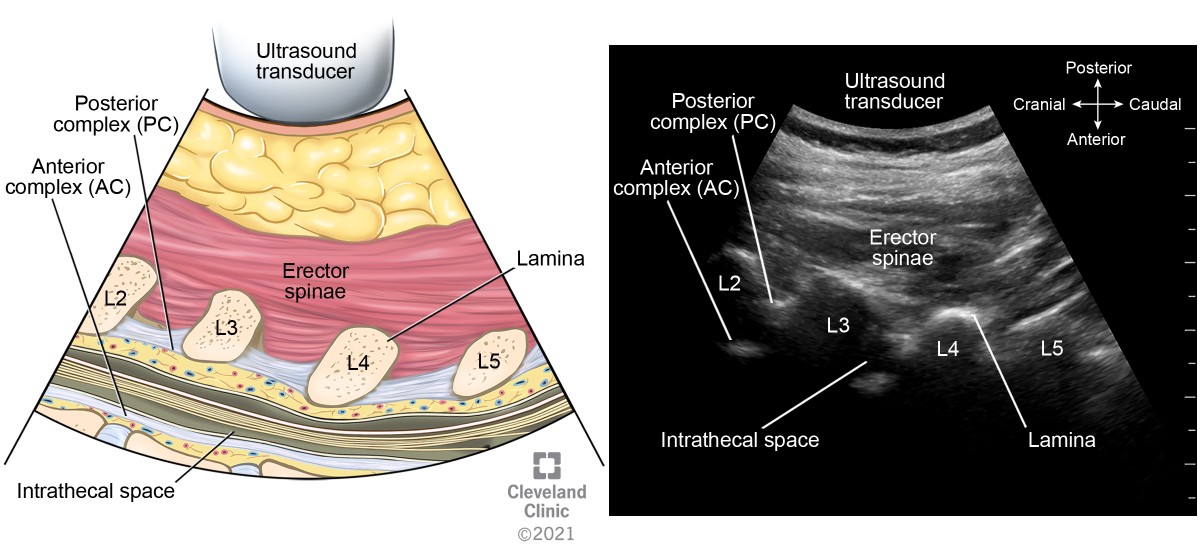
Figure 10. Parasagittal oblique interlaminar view of lumbar spine (sawtooth or horsehead sign)
Transverse Spinous Process View
Once the appropriate interspaces are identified and marked using the parasagittal oblique interlaminar view, rotate the probe 90 degrees and center it on the midline to obtain a transverse spinous process view (Figure 11). The appropriate interspace for a subarachnoid injection should be below L2 to prevent injury to the conus and match the surgical incision for an epidural. The posterior tip of the spinous process is identified superficially as a white hyperechoic line with acoustic shadowing beneath it and a sloping hyperechoic lamina bilaterally (Figure 12). This is the key view for the identification of the midline (Figure 5, transducer position A) and interspinous spaces between the consecutive spinous processes in obese patients.
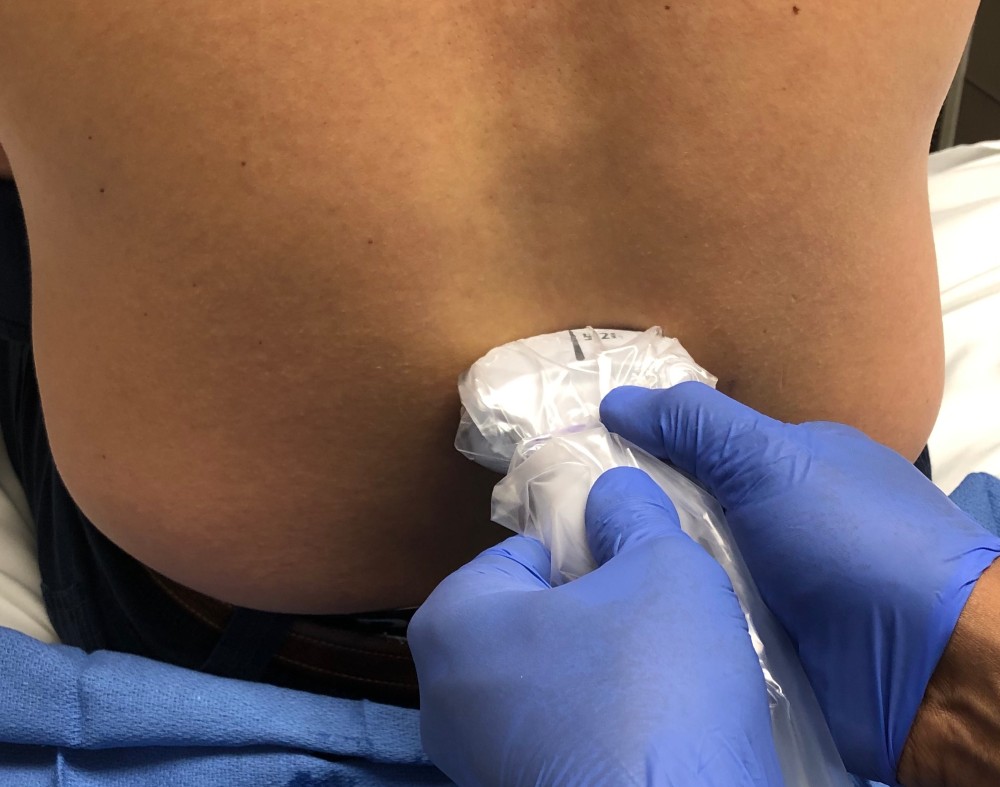
Figure 11. Transducer position for transverse scanning of the lumbar spine
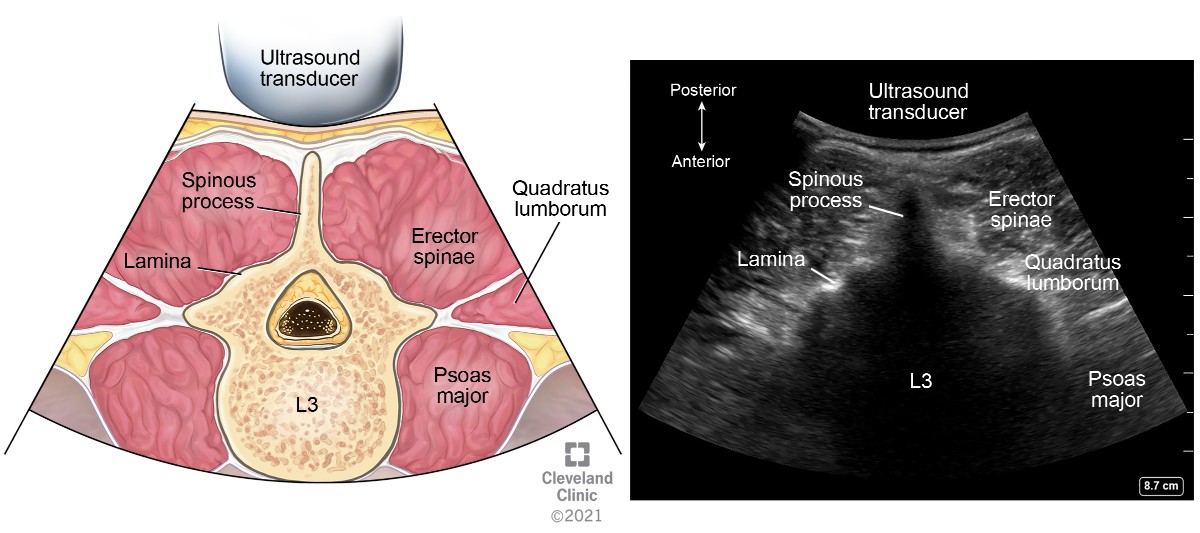
Figure 12. Transverse spinous process view of lumbar spine
Transverse Interspinous View
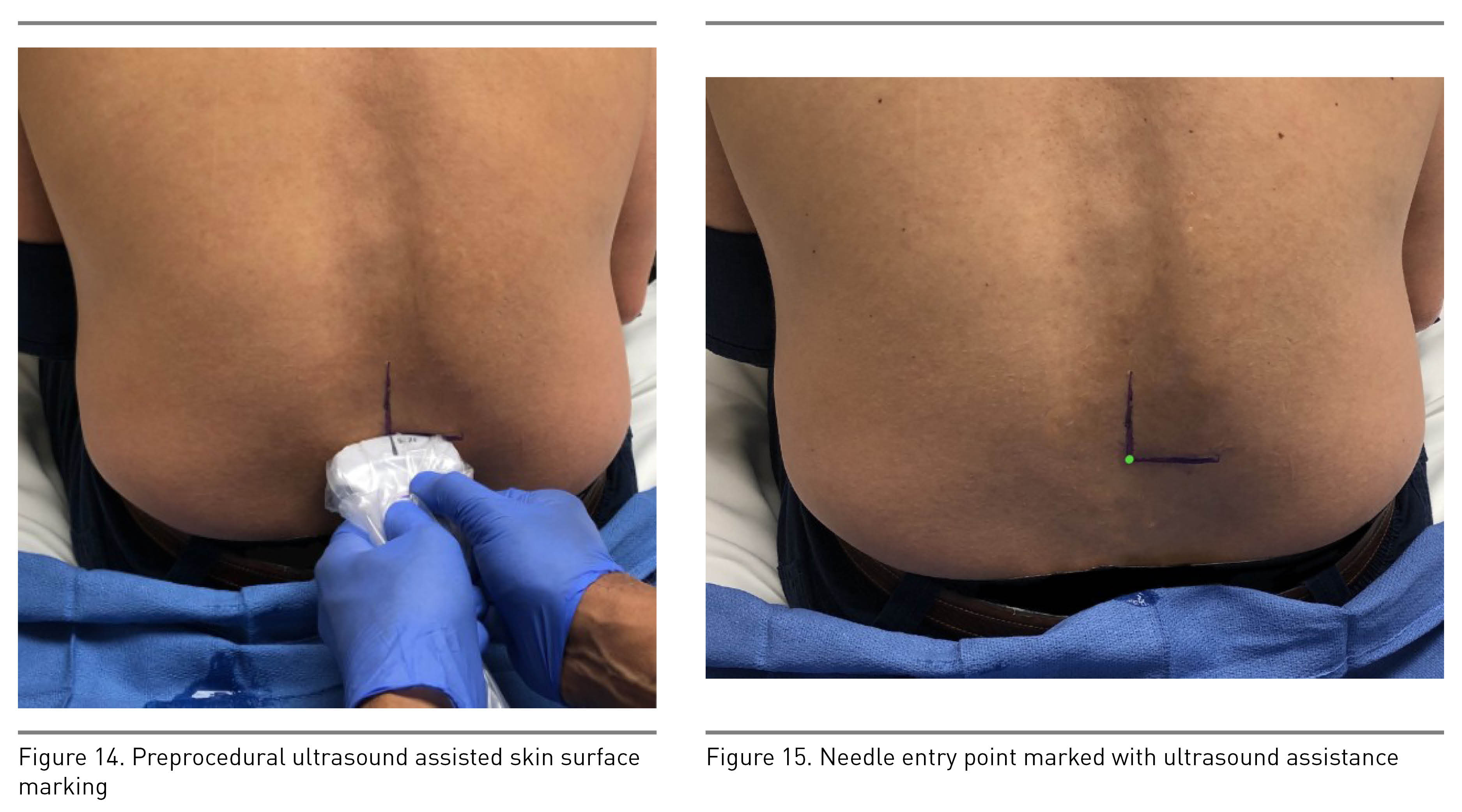
After identification of the spinous process and centering it, move the transducer either cephalad or caudad to the interspinous space (Figure 5 transducer position B). This view, also known as the transverse interlaminar view, allows for visualization of the PC and AC along with articular and transverse processes laterally (Figure 13). The PC’s depth from the skin can be noted in this view and is useful for guiding epidural placement. The transducer may need to be tilted cephalad to visualize both complexes; this angulation can guide the proceduralist’s needle entry for successful placement. After identification of the PC and AC, the skin markings made in horizontal and vertical directions are joined together to mark the needle entry point for neuraxial procedures (Figures 14 and 15). The intrathecal space is evident as hypoechoic space between the PC and AC.
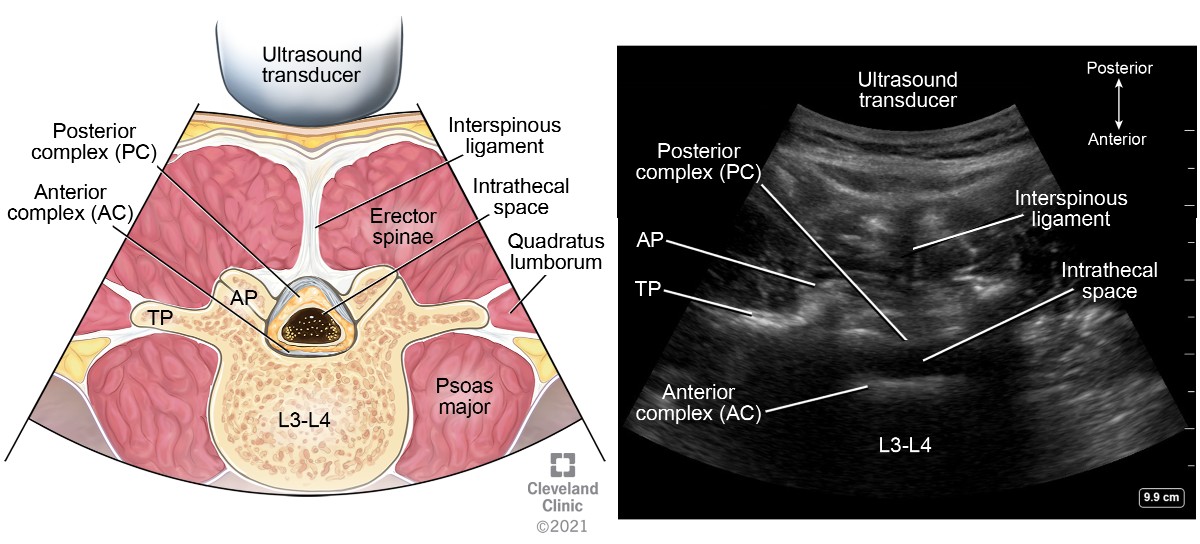
Figure 13. Transverse interspinous view of lumbar spine
AP, articular process; TP, transverse process
Transverse Oblique Foraminal View
From the transverse interspinous view, slide the transducer laterally off the midline in transverse plane to the paraspinal space. Tilt the transducer medially, applying gentle pressure toward the vertebral body. The erector spinae muscle group is identified superficially, with the quadratus lumborum and psoas muscle deeper. The body of the vertebra can be identified along with the superficial nerve root superficial (Figure 16). This view is useful for transforaminal chronic pain interventions, quadratus lumborum, and psoas compartment blocks.
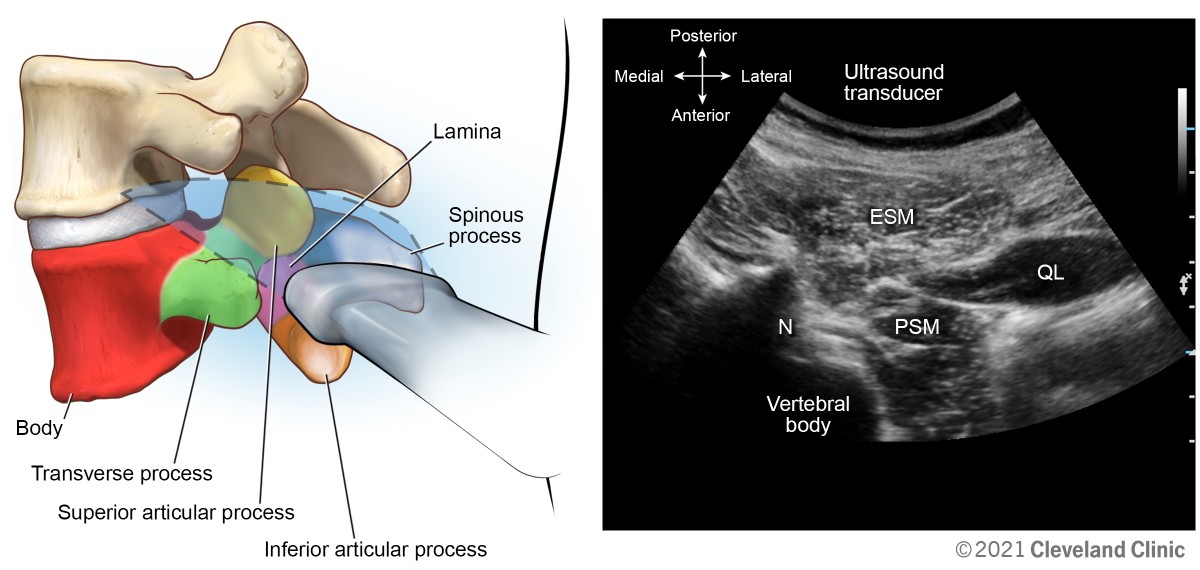
Figure 16. Transverse oblique foraminal view
ESM, erector spinae muscle group; N, lumbar nerve root; PSM, psoas major muscle; QL, quadratus lumborum muscle
Thoracic Spine
The upper (T1–T4) and lower (T9–12) thoracic vertebrae have similar geometry to the cervical and lumbar vertebrae and are amenable for ultrasound scanning (Figure 17). The midthoracic (T5–T8) vertebrae have extreme inferior angulation of spinous process and pose technical challenges for ultrasound scanning.
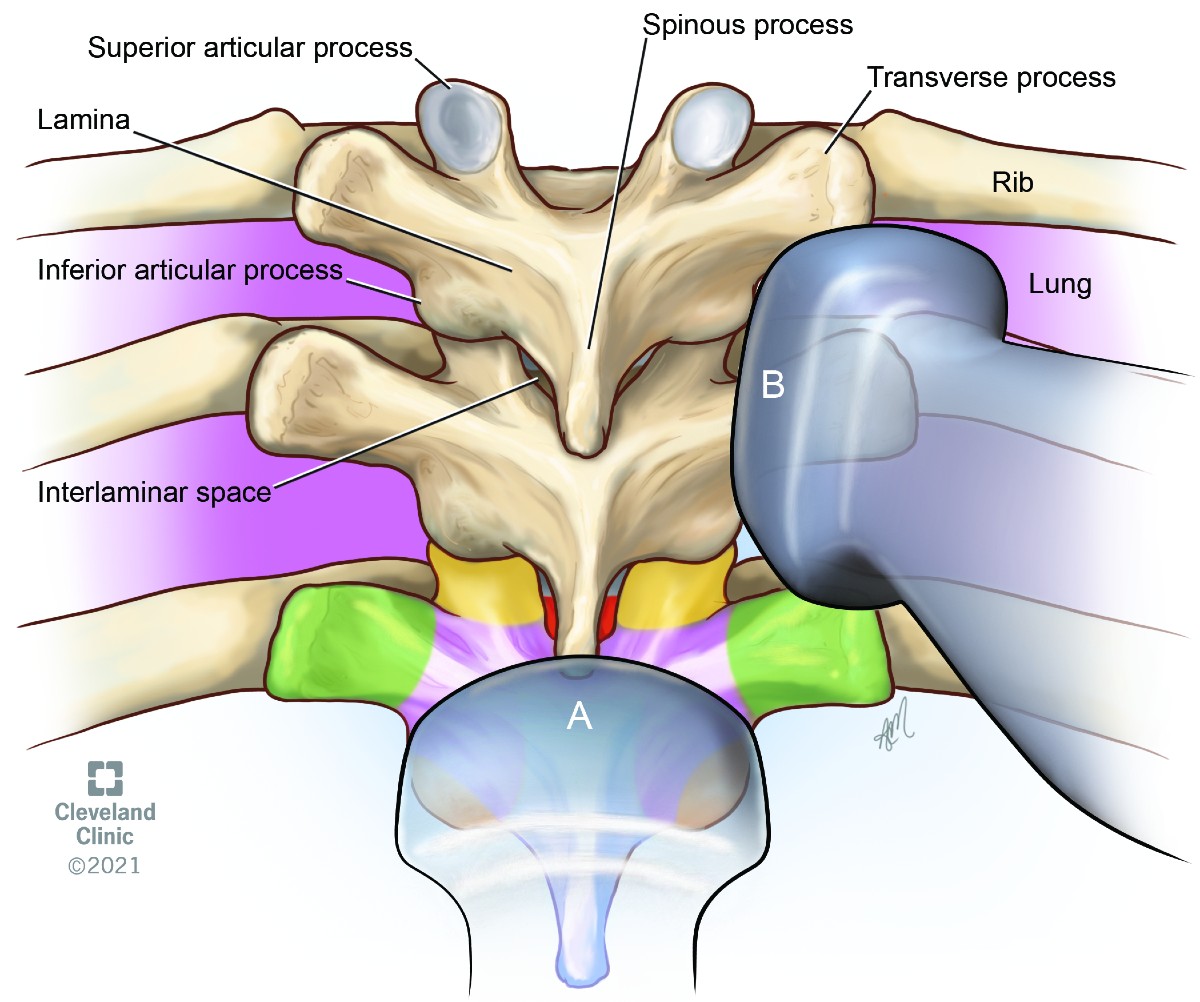
Figure 17. Thoracic vertebra
(A) transverse transducer position
(B) parasagittal oblique transducer position
Parasagittal windows can be obtained by beginning laterally with identification of ribs and pleura, then sliding medially identifying the transverse process, articular process, and lamina. The parasagittal interlaminar view (Figure 18) is used to locate the interlaminar spaces and as a marking point for the neuraxial procedure. The transverse views (Figure 19) are challenging to obtain in the midthoracic spine because the transverse interspinous windows are narrow here. A rib marks the junction of the T12 and L1 vertebra. The 12th rib can be identified to locate the T12 vertebra, and a counting-down approach can be used to locate accurate lumbar intervertebral levels or a counting-up approach for the correct thoracic intervertebral level. Alternatively, the correct level can be determined by counting down from the T1 level, after locating the first rib.
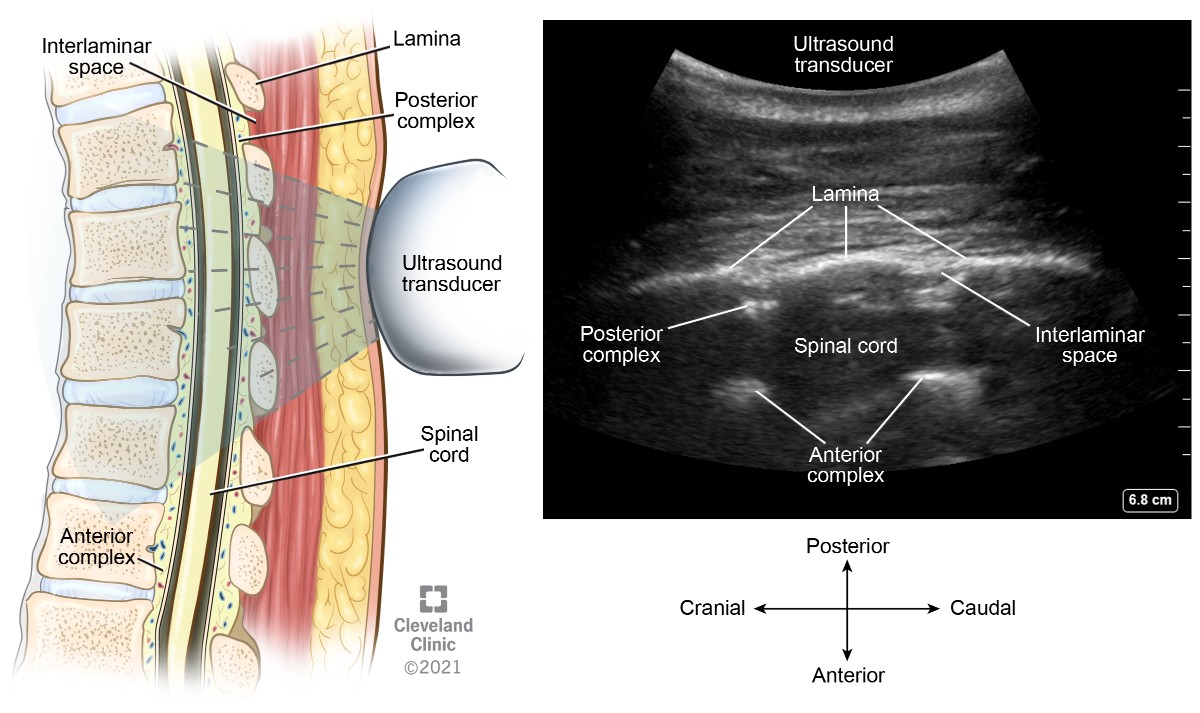
Figure 18. Parasagittal oblique interlaminar view of thoracic spine
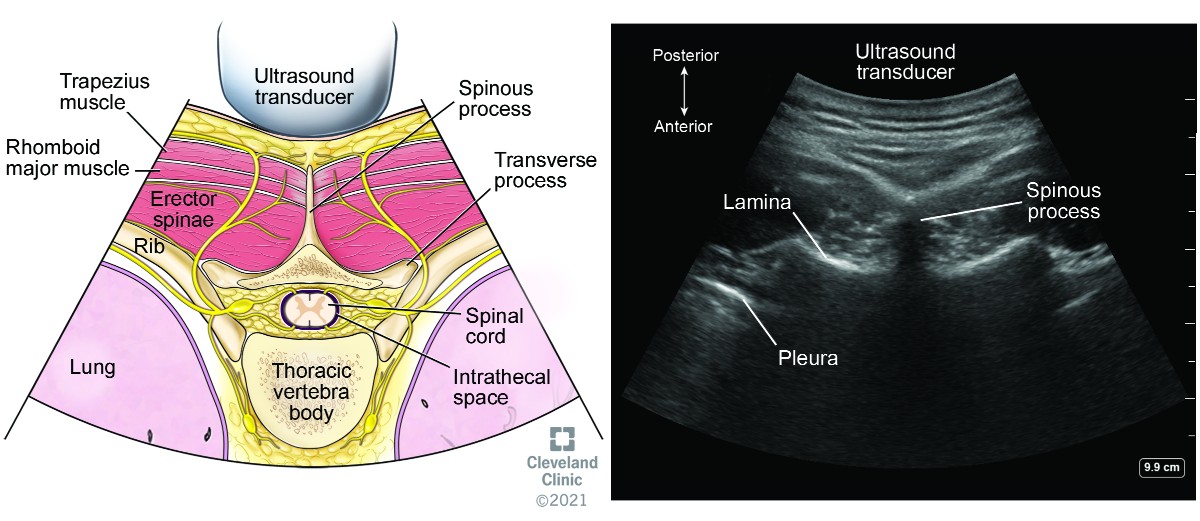
Figure 19. Transverse view of thoracic spine
Scanning Technique
Although multiple variations of the scanning technique have been described in the literature,3,5 the core principles are the same. The authors use the following stepwise approach:
- Position the patient sitting for the procedure. Alternatively, a lateral decubitus position is acceptable.
- Select a curvilinear low-frequency transducer (2–5 MHz) and sterile ultrasound gel as a coupling medium with a sterile probe cover.
- Set the screen depth to 9–11 cm and adjust as needed after initial assessment.
- Start scanning from the parasagittal transverse process view by placing the transducer few centimeters lateral to midline and cephalad to the sacrum. The transverse processes are identified as a trident sign (Figure 7B).
- Slide the transducer medially to obtain a parasagittal articular process view, identified as camel humps (Figure 9).
- Tilt the transducer medially to obtain a parasagittal oblique interlaminar view. Identify the laminae appearing as a sawtooth or horsehead pattern and, subsequently, the PC, AC, and thecal sac (Figure 10).
- Count the interlaminar spaces (acoustic windows) up from the sacrum (Figure 8) in the parasagittal interlaminar view and identify the desired intervertebral level.
- Rotate the transducer 90 degrees to obtain a transverse spinous process view at the desired vertebral level (Figure 12) and make the midline (vertical) marking (Figure 14).
- Obtain the transverse interspinous view by sliding the probe slightly cephalad or caudad. This enables identification of the PC, AC, and thecal sac (Figure 13). Mark the interspaces laterally (Figure 14).
- The intersection of the vertical and transverse skin markings is the needle entry point for ultrasound-assisted neuraxial procedures (Figure 15).
Real-Time Ultrasound Guidance for Neuraxial Procedures
One of the main drawbacks of prepuncture scanning is that during the actual procedure, the anesthesiologist is still operating blind. Additionally, prepuncture scanning provides the operator with measurements at a specific time with the patient in a specific position, but those measurements can change with patient movement, needle insertion, tissue distortion, and needle angle adjustment.8 Active needle tracking during real-time ultrasound allows experienced providers to visualize the needle as it travels the tissue layers, adjusting the trajectory and confirming tip location throughout the procedure.
Studies evaluating the feasibility and success of real-time ultrasound for neuraxial block placement have been promising, and advancements in transducer technology have improved image quality. However, its use as guidance for midline neuraxial blockade is complicated by acoustic shadows from the vertebrae. Additional factors to consider include using a paramedian longitudinal approach, which provides superior quality images compared to classical ultrasound planes used for preprocedural mapping, and using saline as a coupling medium because of the possibility of neurotoxicity with ultrasound gel.9 Currently, real-time ultrasound scanning is a cumbersome and challenging technique with limited clinical utility.
Conclusion
Neuraxial ultrasound imaging should be in the skill set of every anesthesiologist who routinely performs regional anesthesia. Preprocedural imaging minimizes the technical difficulty of spinal and epidural placement with fewer needle passes and skin punctures.10 It helps to accurately identify the midline, vertebral level, and interlaminar space and can predict the depth to the epidural and intrathecal spaces, thereby allowing the anesthesiologist to plan an ideal trajectory for a successful block.11
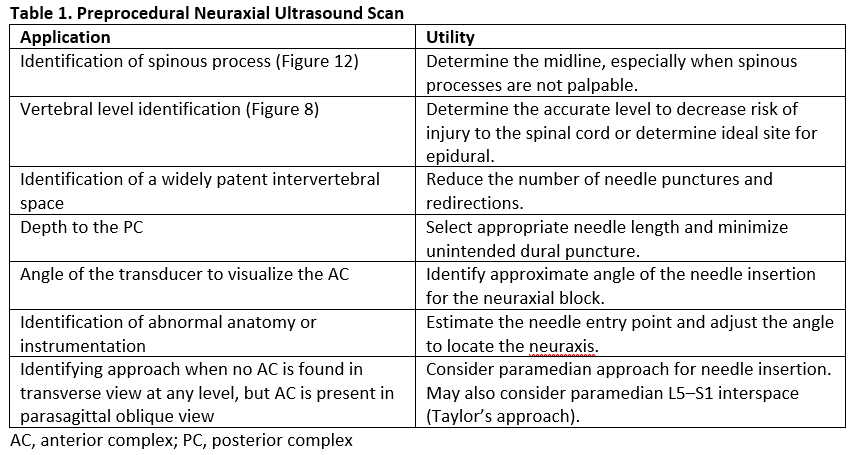
The benefits of preprocedural neuraxial scanning (Table 1) are most noticeable when expert operators conduct the ultrasound examination and when used for patients with predicted difficult spinal anatomy.12 Evidence suggests that preprocedural neuraxial ultrasound imaging may prevent complications such as vascular puncture, headache, and back pain.13 We recommend using preprocedural neuraxial ultrasound imaging routinely to acquire and maintain skills for success with challenging neuraxial procedures and ultrasound-guided paravertebral and paraspinal regional anesthesia blocks.
All images used with permission by Cleveland Clinic, 2021.

Sree Kolli, MD, EDRA, is a clinical assistant professor in the department of General Anesthesia and Acute Pain Management at Cleveland Clinic in Cleveland, OH.

Samer Narouze, MD, PhD, is a clinical professor of Anesthesiology and Pain Medicine at Western Reserve Hospital in Cuyahoga Falls, OH.

Hari Kalagara, MD, FCARCSI, is an assistant professor in the department of Anesthesiology at Mayo Clinic in Jacksonville, FL.
References
- Chin KJ. Recent developments in ultrasound imaging for neuraxial blockade. Curr Opin Anaesthesiol 2018;31(5):608–13. https://doi.org/10.1097/ACO.0000000000000634
- Parate LH, Manjunath B, Tejesh CA, et al. Inaccurate level of intervertebral space estimated by palpation: The ultrasonic revelation. Saudi J Anaesth 2016;10(3):270–5. https://doi.org/10.4103/1658-354X.170104
- Ghosh SM, Madjdpour C, Chin K. Ultrasound-guided lumbar central neuraxial block. BJA Education 2015;16(7):213–20. https://doi.org/10.1093/bjaed/mkv048
- Whitty R, Moore M, Macarthur A. Identification of the lumbar interspinous spaces: palpation versus ultrasound. Anesth Analg 2008;106(2):538–40. https://doi.org/10.1213/ane.0b013e31816069d9
- Chin KJ, Karmakar MK, Peng P. Ultrasonography of the adult thoracic and lumbar spine for central neuraxial blockade. Anesthesiology 2011;114(6):1459–85. https://doi.org/10.1097/ALN.0b013e318210f9f8
- Karmakar MK, Li X, Ho AM, et al. Real-time ultrasound-guided paramedian epidural access: evaluation of a novel in-plane technique. Br J Anaesth 2009;102(6):845–54. https://doi.org/10.1093/bja/aep079
- Provenzano DA, Narouze S. Sonographically guided lumbar spine procedures. J Ultrasound Med 2013;32(7):1109–16. https://doi.org/10.7863/ultra.32.7.1109
- Grau T, Leipold RW, Conradi R, et al. Ultrasound control for presumed difficult epidural puncture. Acta Anaesthesiol Scand 2001;45(6):766–71. https://doi.org/10.1034/j.1399-6576.2001.045006766.x
- Pintaric TS, Hadzic A, Strbenc M, et al. Inflammatory response after injection of aqueous gel into subarachnoid space in piglets. Reg Anesth Pain Med 2013;38(2):100–5. https://doi.org/10.1097/AAP.0b013e318284176a
- Sidiropoulou T, Christodoulaki K, Siristatidis C. Pre-procedural lumbar neuraxial ultrasound—a systematic review of randomized controlled trials and meta-analysis. Healthcare (Basel) 2021;9(4):479. https://doi.org/10.3390/healthcare9040479
- Yoo S, Kim Y, Park SK, et al. Ultrasonography for lumbar neuraxial block. Anesth Pain Med (Seoul) 2020;15(4):397–408. https://doi.org/10.17085/apm.20065
- Elgueta MF, Duong S, Finlayson RJ, et al. Ultrasonography for neuraxial blocks: a review of the evidence. Minerva Anestesiol 2017;83(5):512–23. https://doi.org/10.23736/S0375-9393.16.11650-5
- Schnabel A, Schuster F, Ermert T, et al. Ultrasound guidance for neuraxial analgesia and anesthesia in obstetrics: a quantitative systematic review. Ultraschall Med 2012;33(7):E132–7. https://doi.org/10.1055/s-0029-124572
Leave a commentOrder by
Newest on top Oldest on top