Superior Cluneal Nerve Block: Considerations for Regional Anesthesia and Chronic Pain Management
Pranab Kumar, MD, Ahmad Ghidan, MD, and Gabriel Munoz, MD
Department of Anesthesiology and Pain Medicine
University Hospital Network, Toronto Western Hospital
Toronto, Ontario, Canada
Large epidemiologic studies have shown that up to 85% of axial spine pain is of nonspecific etiology, and 37% of patients with chronic low back pain (LBP) have a neuropathic pain component.1 First described in 1957, superior cluneal nerve entrapment neuropathy (SCN-EN) is a poorly understood clinical entity causing misdiagnosis of LBP.
Anatomy
The SCN derives from the lateral cutaneous branches of the dorsal rami of the T12–L5 spinal nerves and provides sensory innervation to the posterior iliac crest, superior gluteal regions, and sometimes as far as the greater trochanter. SCN branches penetrate the psoas major and paraspinal muscles and traverse through the superficial layer of thoracolumbar fascia before crossing the posterior iliac crest, or they may pass through the gluteal fascia after crossing the iliac crest. The nerve can become entrapped at the thoracolumbar fascia’s penetrating orifice.2
Bogduk et al.1 studied the lumbar dorsal rami and found that the L1–L3 lateral branches pierced the thoracolumbar fascia and became cutaneous, whereas the L4 lateral branch remained entirely intramuscular, and no lateral branch arose from L5. With subsequent cadaveric dissections, Maigne et al.,2 Lu et al.,3 and Xu et al.3 determined that the medial SCN branch consistently passes through the osteofibrous tunnel whereas the intermediate and lateral branches never do. Agur and Dalley4 found that 80% of the medial branches pass through the osteofibrous tunnel, and Yazaki et al.5 reported it at 95%.
Maigne et al.4 found that the SCN passing through the osteofibrous tunnel arises from the L1 nerve roots in 60%, L2 in 27%, and L3 in 13%. In a randomized, combined cadaveric-healthy volunteer trial, Neilson et al.6 showed that SCN branches passing through the osteofibrous tunnel mainly originate from the L3–L5 nerve roots and up to six vertebral levels of origin are involved from the T12–L5 levels. Furthermore, they found that 75% of the dorsal rami of L1, 90% of L2, and 95% of L3 give origin to the SCN, with minor contributions from the dorsal rami of L4 (45%) and L5 (10%).6
Despite those many descriptions and illustrations, the SCN still has just three branches that pass over the iliac crest: medial, intermediate, and lateral. According to Iwanaga et al.,7 55% of medial SCN iliac crests have an osteofibrous tunnel (ie, between the bony groove of the posterior iliac crest and the thoracolumbar fascia). The average distance between that branch (while passing on the iliac crest) and the midline is approximately 7 cm. Kuniya et al.5 concluded that all three SCN branches could travel in a similar tunnel; however, most of the tunnels described are for the medial SCN (see Figure 1A), which thus is the most easily entrapped branch.5
Interestingly, when passing through the well-known osteofibrous tunnel, the medial SCN has multiple tiny branches (variously anastomosed like a small nervous plexus). That thin neural network may serve as a potential pain generator, whether from compression or simply excessive mechanical stress on the neural tissues (eg, tension, twist, or friction under the thoracolumbar fascia) during lumbar spine movements.
Diagnostic Considerations
Maigne et al.’s criteria for SCN-EN are unilateral LBP referred to the iliac crest and buttock, a trigger point over the posterior iliac crest 7 cm from the midline (corresponding to the nerve compression zone), and relief of symptoms by a nerve block.4 It may occasionally produce numbness and radiating pain (Tinel-like signs) in the SCN area during trigger point compression. The Tinel sign is useful for clinical diagnosis; however, a normal sensory examination and bilateral presentation should not exclude an entrapment neuropathy diagnosis.
For diagnostic purposes, the SCN is traditionally blocked with the adapted version of Talu et al.’s technique6 or the more accurate technique involving novel ultrasound injection of local anesthetic. SCN-EN is confirmed when patients experience more than a 75% decrease in symptoms and report a 75% reduction in pain within two hours following the nerve block. Thus, locating the medial SCN can help guide pain management, but electrodiagnostic studies for SCN-EN are unreliable because the nerve is thin and difficult to identify through the skin.
Regional Anesthesia
The novel ultrasound-guided nerve block technique reliably anesthetizes the SCN, which supplies the skin posterior to the area innervated by the subcostal and iliohypogastric nerves. Perioperatively blocking the SCN can significantly increase anesthetic coverage for a variety of hip surgery incisions and may improve analgesia after hip surgery.6,8 When combined with transversalis fascia plane blocks, SCN blocks may also improve analgesia coverage for bone graft harvest from the iliac crest.6 Another application involves injection drug users, who often present for emergency care of a cutaneous abscess in the buttocks following a skin-pop. SCN blocks have been shown effective for analgesia during drainage of upper buttock abscesses.9
SCN Neuropathic Pain
In specific subsets of patients with LBP-related pain and disability, SCN’s role is becoming better understood. Although SCN-EN is relatively rare (1.6%) in Europe, Kuniya et al.8 cited incidence rates as high as 14% in Japanese populations, with approximately 50% of patients with SCN disorders reporting leg symptoms. Trescot10 also described how SCN entrapment produced pseudosciatica pain down the leg, in some cases to the foot, which clinically mimicked radiculopathy with lumbar disc herniation or lumbar spinal canal stenosis. SCN-EN has also been associated with Maigne syndrome and lumbar spine surgeries requiring bone harvest.11 Also, vertebral fractures may exacerbate preexisting SCN entrapment by irritating the nerve at its origin from unstable facet joints or by stretching the SCN with exaggerated kyphosis.
In daily practice, the region produces pain upon palpitation of myofascial trigger points in the gluteus medius, paravertebral muscles, or erector spinae muscle at the attachment site on the posterior iliac crest (ie, iliac crest pain syndrome). In patients with SCN-EN whose pain is not relieved with spinal manipulation, alternatives such as neural mobilization stretches, conservative treatment with repeated SCN blocks, or surgical release of the entrapment under local anesthesia have demonstrated good results.12
Ultrasound-Guided SCN Block
Use of high-resolution ultrasound to identify the SCN has been validated in previous cadaveric studies.1,6,7,13–15 Based on anatomical studies and various scanning protocols,6,16,17 a particular ultrasound scanning window may be used to examine the osteofibrous tunnel.
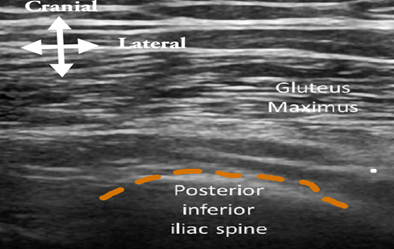
- With the patient in prone position, place the linear transducer (eg, 15-6 MHz) in the transverse plane at the posterior inferior iliac spine (see Figure 1C, position A, and Figure 2A).
- Progressively shift the probe cephalad and laterally (see Figure 1C, position B) until a small fat pad (located among the posterior iliac crest, the thoracolumbar fascia, and the lateral edge of the erector spinae muscle) is optimally visualized. At this point the gluteus maximus muscle disappears and the gluteus medius starts to appear. That anatomical spot has sometimes been referred as the SCN’s “fatty tunnel” (see Figure 2B).
- Redirect the transducer in the long axis of the osteofibrous tunnel so the needle is easily inserted in-plane and the whole tunnel along the course of the thoracolumbar fascia can be dissected (see Figure 1C, position C). Alternatively, use a transverse scan (see Figure 2B) with a lateral to medial in-plane technique. During injection, the erector spinae muscle and posterior layer of thoracolumbar fascia should separate easily.
- Subsequently, if other SCN branches will be blocked, the short-axis view can be helpful in localizing the intermediate and lateral branches (see Figure 2C), which are situated inside the subcutaneous tissue above the gluteus medius muscle.
Figure 1A
| Figure 2A
|
Figure 1B
| Figure 2B
|
Figure 1C
| Figure 2C
|
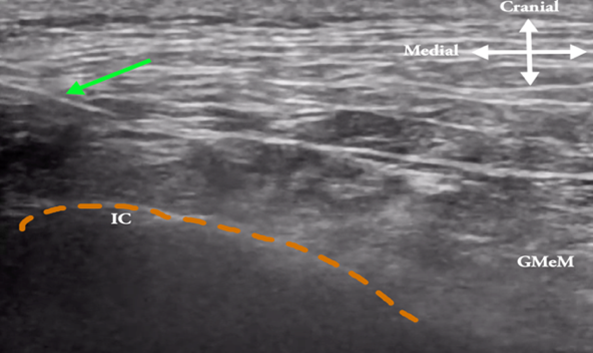
The orange dotted line depicts the ilium crest (IC) border lateral to L1–L5 lumbar vertebrae. The green arrow shows the needle as it penetrates the very lateral part of the posterior layer (green dotted line) of the thoracolumbar fascia. The injectate spreads between the thoracolumbar fascia and erector spinae (ES) muscle. The white arrow denotes in-plane approach to the osteofibrous tunnel and gluteus medius (GMeM) muscle is seen laterally over the Ilium.
Figure 1A: SCN originating mainly from the L1–L3 dorsal rami traversing the iliac crest
Figure 1B: SCN target located around 7cm lateral to the midline of spine over the iliac crest
Figure 1C: When placing the ultrasound probe (green/red rectangle) in a transverse plane over the posterior superior iliac spine (position A), a cranial and lateral shift (black arrow) is performed to visualize the “fat tunnel” (position B) of the SCN’s medial branches (black dotted lines). Position C denotes the longitudinal axis sometimes preferred to enter the osteofibrous tunnel.
Figure 2A: Ultrasound image showing a transverse scan at the posterior superior iliac spine
Figure 2B: Ultrasound image of a transverse scan at 7 cm lateral to the midline over the iliac crest, where the red dot indicates the medial branches of the SCN appearing as multiple black dots inside the fat tunnel bounded by the posterior iliac crest, the thoracolumbar fascia (green dotted line), and the lateral edge of the erector spinae muscle
Figure 2C: Ultrasound image demonstrating a transverse scan at the iliac crest with the needle targeting intermediate and lateral branches
Conclusion
SCN is a potential mediator or generator of both acute and chronic pain. The nerve may be blocked for regional anesthesia in patients undergoing hip surgery or iliac crest bone graft harvest. SCN’s potential role in chronic pain conditions should be considered during the diagnostic process to minimize the risk of false diagnosis because of overlapping patterns for LBP. Ultrasound-guided SCN blocks may serve as a useful diagnostic test and can be routinely employed for targeted steroid injections. With more studies, advanced therapies like cryoablation and peripheral neuromodulation may become promising, minimally invasive techniques for the treatment of neuropathy-mediated SCN pain.
References
1. Bogduk N, Wilson AS, Tynan W. The human lumbar dorsal rami. J Anat. 1982;134(Pt 2):383–97. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1167925
Iwanaga J, Simonds E, Schumacher M, Yilmaz E, Altafulla J, Tubbs RS. Anatomic study of the superior cluneal nerve and its related groove on the iliac crest. World Neurosurg. 2019;125:e925–8. https://doi.org/10.1016/j.wneu.2019.01.210
Herring A, Price DD, Nagdev A, Simon B. Superior cluneal nerve block for treatment of buttock abscesses in the emergency department. J Emerg Med. 2010;39(1):83–5. https://doi.org/10.1016/j.jemermed.2009.08.033
.jpg?sfvrsn=d894d83a_1)
.jpg?sfvrsn=6f71ec0c_1)
.jpg?sfvrsn=b051a84c_1)
.jpg?sfvrsn=86427b45_1)