How I Do It
Learn from your colleagues. ASRA Pain Medicine members share how they perform common procedures and accomplish other tasks.
- Listed by publication date, most recent on top
How I Do It: Magic Potion for Spinal Anesthesia
Cite as: Hartmann P. How I do it: magic potion for spinal anesthesia. ASRA News. 2021;46. https://doi.org/10.52211/asra050121.030.
The benefits of spinal anesthesia for lower-extremity joint replacement have been well described in recent literature.1 The popularity of joint replacements over the past decade have also, predictably, resulted in more revision arthroplasties and subsequent increases in surgical complexity. This increased surgical complexity often requires significant prolongations in surgical and anesthesia times. The clinical outcome improvements associated with the provision of neuraxial anesthesia for these surgical procedures require an adjustment from usual neuraxial medication administration to ensure that adequate anesthesia time is provided.2 We have developed a single-injection intrathecal technique by adjusting medication and dose to meet this challenge.
How I Do It
The patient is admitted and has an IV inserted in our dedicated regional nerve block preoperative preparation and procedure area. Patients are greeted by a member of the anesthesia care team, and, following a discussion of the procedure and expected benefits and risks, written informed consent is obtained. Standard ASA monitors are placed, and sedation may be provided via the administration of low-dose midazolam. The patient is assisted into the seated position, and a curvilinear ultrasound probe is used for preprocedure marking. The benefits of ultrasound guidance have been well described in recent literature, and it is now a standard part of our practice.3 The sacrum is identified in the paramedian sagittal oblique view as a long hyperechoic line with a cephalad drop off to the posterior longitudinal ligament of the L5-S1 space (Figure 1). The location of this space is marked on the skin. The visualization of the longitudinal ligament confirms that there is an open needle path to the intrathecal space at this location. The ultrasound transducer is then turned 90° to visualize the transverse plane. The L5 spinous process is identified as a brightly hyperechoic, superficial structure immediately cephalad to our marked L5-S1 interspace (Figure 2). The location of the spinous process is marked at the skin, indicating the location of midline at that level. The site is prepped and draped in a sterile fashion. A 25-gauge non-cutting spinal needle is used to access the intrathecal space via a midline approach at either the L4-5 or L5-S1 interspace. We use either a Sprotte or Whitacre 25-gauge needle. If difficulty is encountered, a 22-gauge Sprotte needle is used for a para-median approach at the L5-S1 space. This approach is visualized with the paramedian sagittal oblique ultrasound view and offers the best chance for success. Once the intrathecal space is accessed, as demonstrated by a continuous return of cerebrospinal fluid, our local anesthetic of choice can be injected.
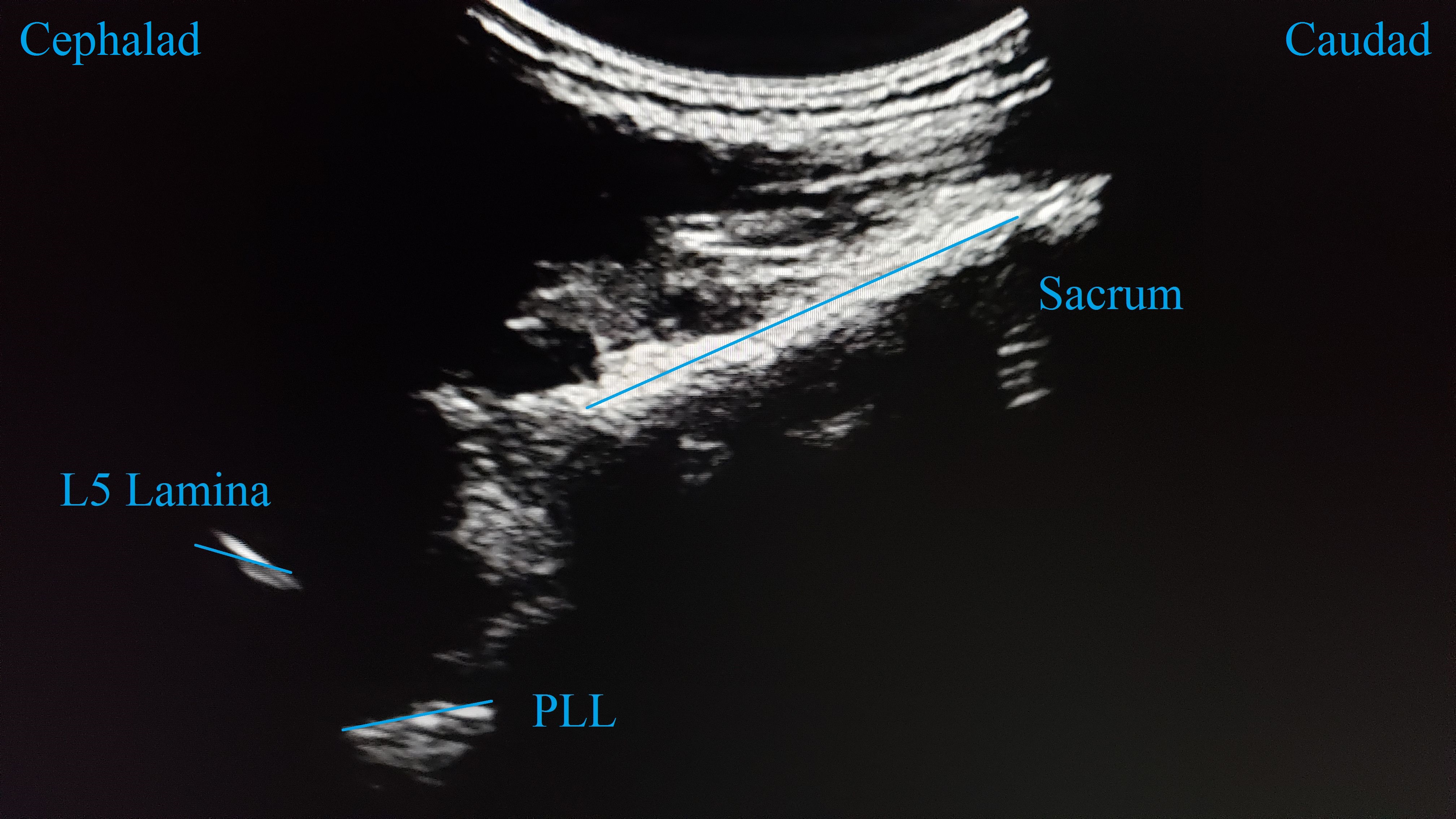
Figure 1. Paramedian sagittal oblique view of the L5-S1 interspace and posterior longitudinal ligament (PLL).
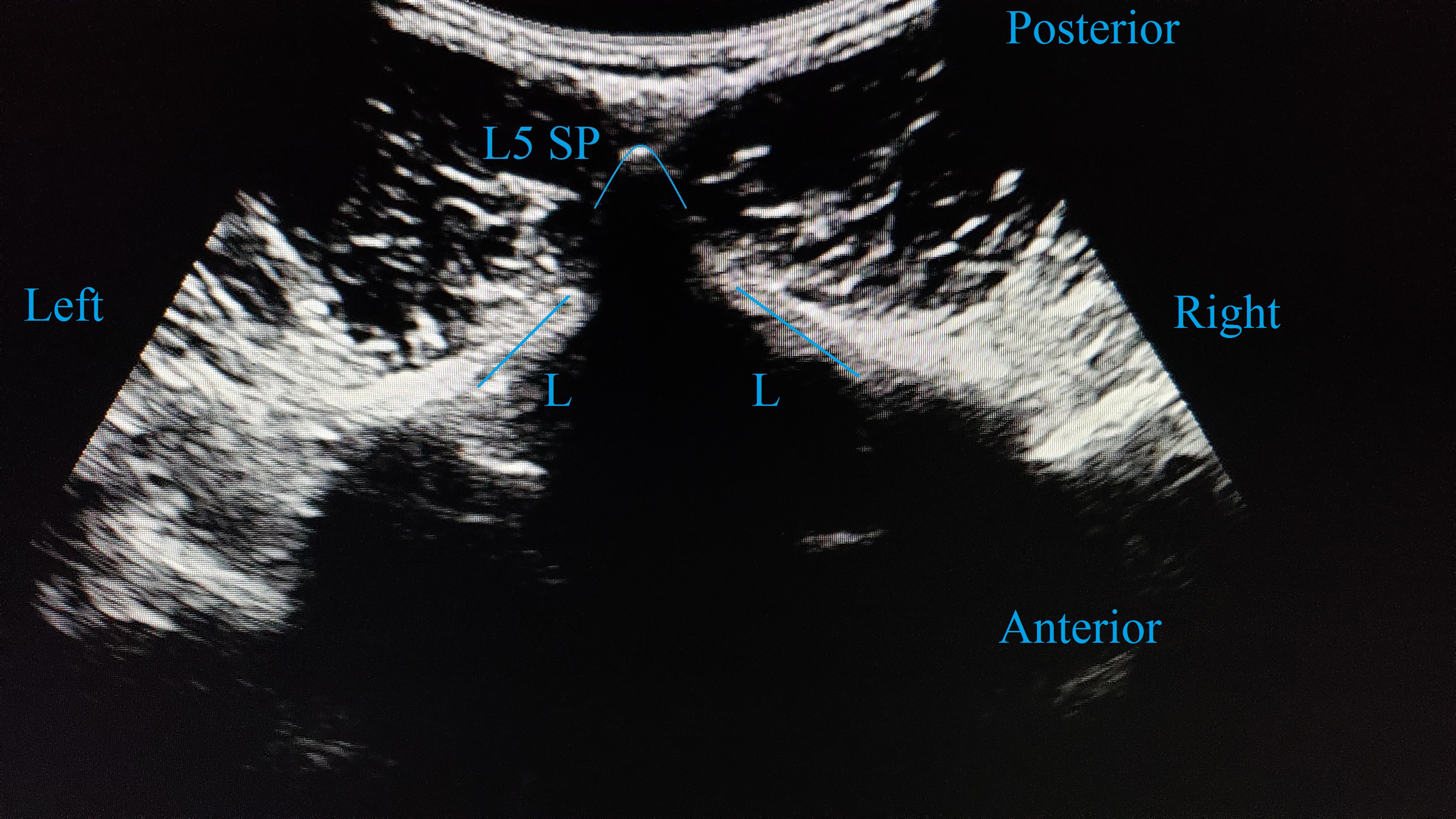
Figure 2. Transverse midline view of L5 spinous process (SP) and lamina (L).
Intrathecal medication and dose are selected based on best estimates for case duration (Table 1). For most primary joint replacements, 2 mL of 0.75% bupivacaine in dextrose is used. For patients being discharged on the same day, either 2-2.5 mL of 2% mepivacaine or 1-1.5 mL of 0.75% bupivacaine with dextrose are utilized to encourage faster block resolution.
Table 1. Surgical procedure examples with local anesthetic and dose.
| Procedure | Intrathecal Local | Duration of Surgical Block, Hours |
| Ambulatory joint | 2% mepivacaine 40 mg or 0.75% bupivacaine with dextrose 7.5-10 mg | 1-2 |
| Primary joint | 0.75% bupivacaine with dextrose 15 mg | 2 |
| Revision joint | 0.5% isobaric bupivacaine 15-20 mg | 3-4 |
| Complex revision | 0.5% isobaric bupivacaine 25 mg | 4-5 |
| Sequential bilateral joints | 0.5% isobaric bupivacaine 25 mg with 30 mcg clonidine | 6-8 |
For revision joint replacements, 0.5% isobaric bupivacaine is used at variable doses depending on potential case length. For an uncomplicated revision, 3.5-4 mL of 0.5% isobaric bupivacaine (17.5-20 mg) will reliably produce a surgical block for 3-4 hours. For potentially longer procedures, 5 mL of 0.5% isobaric bupivacaine (25 mg) will yield a surgical block for 4-6 hours. In our experience, the dermatomal spread is not appreciably increased at these doses. In the rare circumstance where a surgical block of an even longer duration is required, the addition of clonidine (30-50 mcg) can prolong this block to the 6- to 8-hour range. Clonidine has been shown to prolong both analgesia and motor block, but with an increased incidence of hypotension.4 While intrathecal additives have been frequently investigated, we are not familiar with any studies regarding the use of higher doses of intrathecal isobaric bupivacaine.
After delivery of the desired intrathecal medication, the patient is positioned supine. They are monitored for several minutes for any hemodynamic changes before being transported to the operating room. The patient is then comfortably positioned on the operating table. Upon patient preference, moderate sedation with either propofol or dexmedetomidine infusion is initiated, taking care to monitor for airway obstruction. Patients receiving bupivacaine doses in the 20-25 mg range, and especially patients who also received clonidine additive, may require a low-dose infusion of a vasopressor to support their blood pressure.
We have had no incidences of high spinal blockade with this approach in our orthopedic population. Members of our group have been using this dosing practice for over 15 years and have performed more than 100 spinal blocks with bupivacaine doses of 20 mg or higher. It is important to consider that our injection site is confirmed with ultrasound to be either L4-5 or L5-S1 and that high-dose isobaric bupivacaine is being used. Higher injection site and the use of 0.75% bupivacaine with dextrose may increase this risk significantly depending on patient positioning immediately following blockade. In addition, this method is not used in the obstetric population. Standard contraindications to neuraxial anesthesia are observed, but we have not identified any additional contraindications specific to the higher intrathecal local anesthetic dosing. This practice also requires clear communication with the post-anesthesia recovery team, as the long block duration can necessitate a longer time in recovery.
Our approach offers a streamlined and reliable process for joint arthroplasty under neuraxial anesthesia utilizing familiar medications while avoiding potential technical and reliability issues of the combined spinal-epidural technique. Extended duration of surgery does not have to be an absolute contraindication for single-injection spinal anesthesia. From a simple primary arthroplasty to a complicated joint revision, we have a magic potion to get the job done.
Patrick Hartmann, MD, is an instructor of anesthesia and the director of orthopedic anesthesia at Dartmouth-Hitchcock Medical Center in Hanover, NH.
References
- Turcotte JJ, Stone AH, Gilmor RJ, Formica JW, King PJ. The effect of neuraxial anesthesia on postoperative outcomes in total joint arthroplasty with rapid recovery protocols. J Arthroplasty. 2020;35(4):950-954. https://doi.org/10.1016/j.arth.2019.11.037.
- Wilson JM, Farley KX, Erens GA, Guild GN 3rd. General vs Spinal Anesthesia for Revision Total Knee Arthroplasty: Do Complication Rates Differ?. J Arthroplasty. 2019;34(7):1417-1422. https://doi.org/10.1016/j.arth.2019.03.048.
- Perlas A, Chaparro LE, Chin KJ. Lumbar neuraxial ultrasound for spinal and epidural anesthesia: a systematic review and meta-analysis. Reg Anesth Pain Med. 2016;41(2):251-260. https://doi.org/10.1097/AAP.0000000000000184.
- Elia N, Culebras X, Mazza C, Schiffer E, Tramèr MR. Clonidine as an adjuvant to intrathecal local anesthetics for surgery: systematic review of randomized trials. Reg Anesth Pain Med. 2008;33(2):159-167. https://doi.org/10.1016/j.rapm.2007.10.008.
Leave a commentOrder by
Newest on top Oldest on top