How I Do It
Learn from your colleagues. ASRA Pain Medicine members share how they perform common procedures and accomplish other tasks.
- Listed by publication date, most recent on top
How I Do It: Stellate Ganglion Blocks for Refractory Ventricular Electrical Storm
Cite as: Condrey J, George R, Wilson S. How I do it: stellate ganglion blocks for refractory ventricular electrical storm. ASRA Pain Medicine News 2022;47. https://doi.org/10.52211/asra110122.046
Refractory ventricular arrhythmias, such as ventricular fibrillation (VF) and ventricular tachycardia (VT), are challenging clinical entities to treat. The recurrence of three or more episodes of VT/VF within a 24-hour period is known as ventricular electrical storm (VES).1 Associated with increased morbidity, VES is most commonly encountered in the surgical and cardiac intensive care units (ICUs). The mechanism behind VES is thought to be a combination of several factors and usually presents in patients with some combination of structural heart disease, an inciting trigger, and activation of sympathetic activity.1
The goal of treating VES is to terminate the malignant arrhythmia. Treatment is complex, requiring a multidisciplinary, multimodal approach. Pharmacologic therapy may include beta blockers, anti-arrhythmic drugs, mechanical circulatory support, reprogramming of implanted cardiac devices, heavy sedation, and mechanical ventilation.1 Despite maximal medical therapy, episodes of VT/VF sometimes persist. Neuromodulation, the reduction of the efferent sympathetic tone to the myocardium, may help suppress these refractory arrhythmias. Neuromodulation can be achieved with either a high thoracic epidural or a stellate ganglion block (SGB).2,3,4 As patients with VES are often anticoagulated or have other contraindications to placement of a thoracic epidural, SGB is an attractive option for reduction in sympathetic tone in this patient population. An SGB via single injection or catheter may help bridge the patient to a more definitive therapy, such as catheter ablation, surgical sympathectomy, or even heart transplant. The regional anesthesiologist may be consulted for placement of an SGB to assist in management of patients with VES.
The recurrence of three or more episodes of VT/VF within a 24-hour period is known as ventricular electrical storm (VES).
The stellate ganglion is a bilateral structure that is formed from the inferior cervical and first thoracic sympathetic ganglia. It is usually located at the level of C7 or T1 between the longus colli muscle and carotid artery, lateral to the vertebral body. The stellate ganglion provides efferent sympathetic outflow to the myocardium, upper extremity, neck, and face. It is important to note that the left stellate ganglion contributes more to the sympathetic tone of the myocardium and, for this reason, a left SGB is preferred for the indication of VES.5 There are many vital structures in the vicinity of the stellate ganglion, such as the carotid artery, internal jugular vein, thyroid gland, vertebral artery and vein, brachial plexus, and the trachea. Careful continuous needle visualization is required to avoid injury to one of these structures. Despite the proximity of the stellate ganglion to large vascular and neural structures, the technical performance of the SGB is similar to other regional anesthetic techniques. Thus, the skill set of the regional anesthesiologist is uniquely suited to SGB placement.
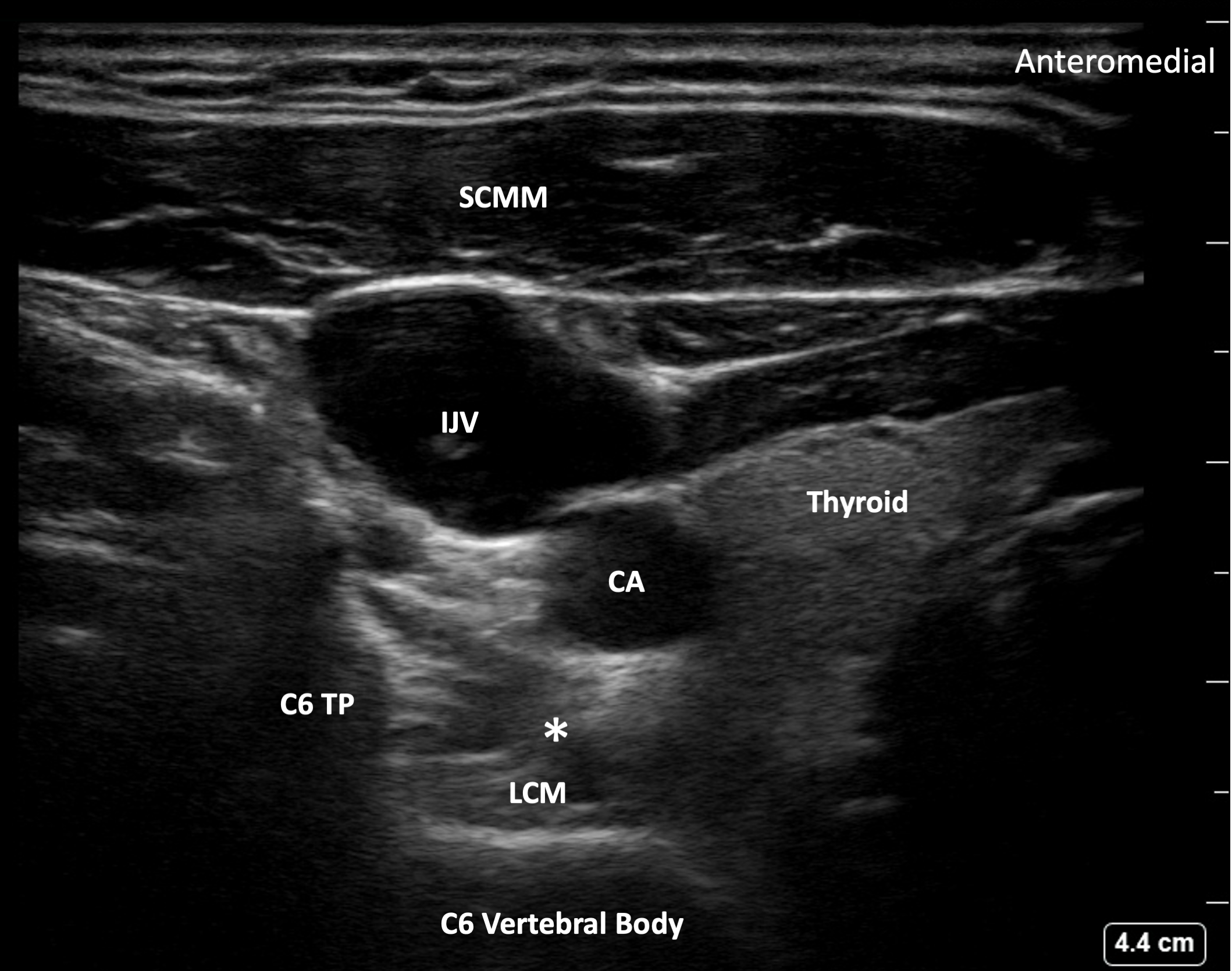
In our institution, the critical care team consults the regional anesthesia team when they have a patient in VES who may benefit from SGB placement. Performance of the SGB requires care but is relatively straightforward. The block is performed with the patient in the supine position, and, for reasons described above, blockade of the left stellate ganglion is preferred. The patient’s head should be turned to the right side, and the ultrasound placed opposite the anesthesiologist performing the block in an unobstructed area to provide ease of visualization. The left side of the neck should be prepped and draped in the usual sterile fashion. A high frequency (6-15 MHz) linear array ultrasound probe is used to identify the target area at the level of C6 by identifying the C6 nerve root of the brachial plexus via a transverse orientation, similar to the sonography of an interscalene block. Once the interscalene view has been identified, the C7 root can be visualized along with the vertebral artery (using color doppler if needed) at the inferior portion of the screen. From here, the linear probe should be advanced in a cephalad fashion until the vertebral artery is no longer seen and the characteristic shape of the C6 transverse process comes into view. With the ultrasound placed on the anterolateral surface of the neck in a transverse orientation, the stellate ganglion lies deep to the anterior scalene muscle and carotid artery, superficial to the longus colli muscle, and anteromedial to the C7 or T1 transverse process. While planning the path of the needle, we recommend turning on color doppler to verify that no vessels lie in the trajectory.
The stellate ganglion is usually located at C7 or T1, but injecting local anesthetic at the level of C6 will result in caudad spread to the stellate ganglion. The vertebral artery and vein exit the boney protection of the vertebral foramen at the level of C7 and, as a result, injection below C6 may carry a greater risk of vascular puncture and should be avoided. Once the target area has been visualized and a needle trajectory planned, the needle is advanced utilizing an in-plane approach in a lateral to medial fashion with constant visualization (Figure 1).6 After careful aspiration, we inject 10 cc of ropivacaine 0.25%. A low concentration and low volume of local anesthetic is used for the block to reduce the risk of local anesthetic systemic toxicity (LAST), as these patients may already be on a lidocaine infusion for suppression of ventricular arrhythmias. At our institution, we favor placing a catheter after the initial bolus to continue local anesthetic delivery and provide prolonged suppression of VES, while the ICU team, cardiology, and other teams formulate a plan for more definitive management. Continuous local anesthetic infusion with catheter placement may reduce the need for repeat SGB, and catheters may be used to bolus local anesthetic should VES recur. After the initial ropivacaine bolus, we thread a catheter and initiate an infusion of ropivacaine 0.2% at a rate of 5 ml/h alone or 1 ml/h (for catheter patency) with a programmed intermittent bolus of 10 ml every two hours. While our team does not advocate for prolonged peripheral nerve block catheters (greater than 7 days), the unique nature of these patients necessitates discussion between the managing teams on length of catheter continuation based on treatment plan.
Figure 1. Target image for the SGB. TP – Transverse Process. SCMM – Sternocleidomastoid muscle. IJV – Internal Jugular Vein. CA- Carotid Artery. LCM – Longus Colli Muscle. Asterisk - represents the needle endpoint.
Adverse effects of SGB can be mitigated with proper attention to detail. As mentioned above, excellent needle visualization is necessary to avoid injury to any of the neurovascular structures in the vicinity of the stellate ganglion, especially as patients with VES are frequently anticoagulated. Phrenic and recurrent laryngeal nerve paresis can occur with the SGB, and bilateral SGBs should be avoided unless the patient is already intubated and a left-sided SGB has failed to achieve arrhythmia suppression. Development of Horner’s syndrome with ipsilateral ptosis, miosis, and anhidrosis due to sympathectomy from the block is indicative of a working block; however, Horner’s syndrome can be nonspecific and can also be difficult to observe in sedated, intubated ICU patients.5 Hematoma is a potential risk; although, we have not experienced this at our institution even with patients usually receiving anticoagulation. Finally, aspiration prior to any injection of local anesthetic is crucial, as even small amounts of local anesthetic injected into the vertebral artery may result in neurologic symptoms of local anesthetic systemic toxicity, such as seizures.
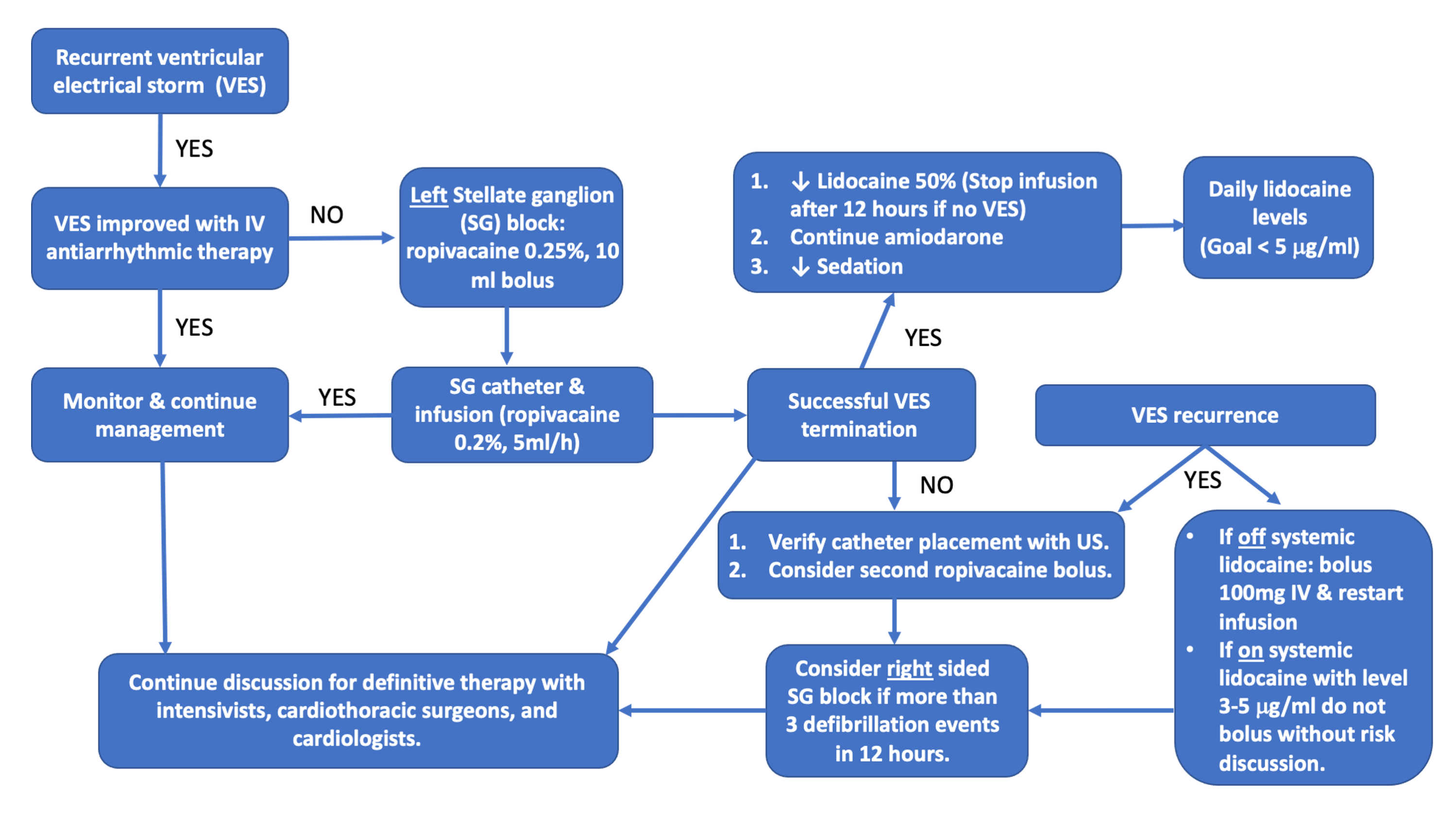
Coordination between care teams is essential. Patients are typically in the ICU with intensivists, cardiothoracic surgeons, and cardiologists collaborating on the patient’s care. We have developed an SGB management pathway at our institution, primarily for the purpose of reducing risk of LAST in patients who are receiving concurrent lidocaine infusions and to manage ventricular arrhythmias should they recur (Figure 2).
Figure 2. Management diagram for stellate ganglion block.
In conclusion, SGB is a valuable addition to the regional anesthesiologist’s armamentarium and can serve as a bridge to more definitive therapy for refractory ventricular arrhythmias.
Jackson M. Condrey, MD, is an assistant professor in the department of anesthesia and perioperative medicine at the Medical University of South Carolina in Charleston.
Renuka George, MD, is an associate professor in the department of anesthesia and perioperative medicine at the Medical University of South Carolina in Charleston.
Sylvia H. Wilson, MD, is a professor and the chief of the division of regional anesthesia and acute pain medicine in the department of anesthesia and perioperative medicine at the Medical University of South Carolina in Charleston.
References
- Geraghty L, Santangeli P, Tedrow UB, et al. Contemporary management of electrical storm. Heart Lung Circ 2019;28(1):123-33. https://www.doi.org/10.1016/j.hlc.2018.10.005
- Martinek M, Manninger M, Schönbauer R, et al. Expert consensus on acute management of ventricular arrhythmias - VT network Austria. Int J Cardiol Heart Vasc 2021;34:100760. https://www.doi.org/10.1016/j.ijcha.2021.100760
- Meng L, Tseng CH, Shivkumar K, et al. Efficacy of stellate ganglion blockade in managing electrical storm: a systematic review. JACC Clin Electrophysiol 2017;3(9):942-9. https://www.doi.org/10.1016/j.jacep.2017.06.006
- Rajesh MC, Deepa KV, Ramdas EK. Stellate ganglion block as rescue therapy in refractory ventricular tachycardia. Anesth Essays Res 2017;11(1):266-7. https://www.doi.org/10.4103/0259-1162.194566
- Ganesh A, Qadri YJ, Boortz-Marx RL, et al. Stellate ganglion blockade: an intervention for the management of ventricular arrhythmias. Curr Hypertens Rep 2020;22(12):100. https://www.doi.org/10.1007/s11906-020-01111-8
- Narouze S. Ultrasound-guided stellate ganglion block: safety and efficacy. Curr Pain Headache Rep 2014 Jun;18(6):424. https://www.doi.org/10.1007/s11916-014-0424-5



Leave a commentOrder by
Newest on top Oldest on top