How I Do It: Perioperative Analgesia for Lower Limb Amputations
Cite as: Do DA., Pozek JJ. How I do it: perioperative analgesia for lower limb amputations. ASRA News. 2021;46. https://doi.org/10.52211/asra050121.031.
Lower-limb amputation surgery is a major life-changing procedure that can result in significant morbidity and mortality. The incidence of these procedures is anticipated to increase in the United States with rising rates of associated illnesses (ie, diabetes, vascular disease, etc).1 The unique patient population that receives lower limb amputation tends to have multiple comorbidities. Perioperative pain following lower extremity amputation can be difficult to manage, increasing the risk for prolonged hospitalization due to uncontrolled pain and development of chronic pain after amputation (CPAP). Subsequently, these patients can become a long-term burden on the medical system if they are not managed appropriately with a multidisciplinary analgesic approach in the perioperative period.
It is critical to identify patients who are at high risk for the development of CPAP.
Severe acute postoperative pain has been associated with an increased risk of CPAP.2 Previous research has demonstrated that 50% to 85% of amputees develop phantom limb pain that, once established, may be difficult to manage.3 Implementation of a perioperative pain management guideline has demonstrated the ability to improve pain scores and reduce opioid consumption in patients undergoing lower-limb amputations.4 This discussion will outline our institution’s treatment regimen for patients undergoing lower-limb amputation.
Preoperative Setting
Prior to surgery, it is critical to identify patients who are at high risk for the development of CPAP and work to coordinate and optimize pre-existing conditions associated with poor pain-related outcomes. For example, patients with a history of multiple amputations, psychiatric illnesses, and preoperative pain with baseline opioid requirements may benefit from optimization efforts.2
A thorough patient history and physical examination are necessary to stratify risk factors for developing chronic pain after amputation. Multiple psychiatric illnesses have been associated with a higher risk of developing CPAP. Anesthetic management for these patients begins with proper psychological evaluation to identify and engage support systems, allay any apprehension, evaluate current coping mechanism strategies, and undergo training to develop coping strategies that might be helpful following surgery. Preoperative anxiety and poor socioeconomic support also correlate with an increased risk for developing chronic pain. We use this time to educate the patient on expectations regarding pain, the planned analgesic regimen, and anticipated events while providing reassurance and additional resources.
Another crucial aspect is preoperative optimization of the analgesic regimen before lower-limb amputation to preemptively reduce noxious sensitization of the central nervous system. Karanikolas et al. demonstrated that an analgesic regimen started 48 hours prior to surgery decreased phantom limb pain at 6 months.5 While it is not possible to identify every patient for lower-limb amputation 48 hours prior to surgery, we work with our surgeons to identify these patients and implement our analgesic plan as soon as possible.
Once we have identified a patient for lower-limb amputation, we begin an aggressive multimodal analgesic regimen that includes early regional anesthesia and continuing chronic opioid therapy. Abruptly stopping the patient’s baseline opioid demand can result in inadequate pain control and increase the risk of acute withdrawal.6
Multiple non-opioid adjuncts must be utilized, since on a neurochemical level, pain is modulated through multiple receptors. For lower-limb amputation, our multimodal approach to preemptive analgesia includes gabapentin, acetaminophen, NSAIDs, and an appropriate regional anesthetic technique.
Regional Technique: Below Knee Amputation
Regional anesthesia for below-knee amputation (BKA) is an important analgesic modality. Preoperative regional anesthesia allows attenuation of the surgical stress response and decreases intraoperative opioid requirements. Waiting until after the noxious stimulus and the development of pain in the postoperative period can heighten central pain sensitization and contribute to the development of chronic postoperative pain.7 In our institution, we have found that timing regional anesthesia interventions prior to surgery has been crucial to program success.
Our ideal regional anesthetic technique for BKA consists of the placement of dual peripheral nerve catheters (PNC): popliteal sciatic (Figure 1) and saphenous (Figure 2). The sensory innervation of the lower extremity below the knee mainly consists of the sciatic and saphenous nerve, and we believe blockade of both nerves is important in an ideal anesthetic plan for BKA. The advantage of using PNCs instead of the single-shot regional approach is that it allows for a longer duration of analgesia into the postoperative period. In addition, placement of PNCs allows for the scheduling of this procedure to occur at a time convenient for the regional anesthesia team in a manner that decreases operating room delays.
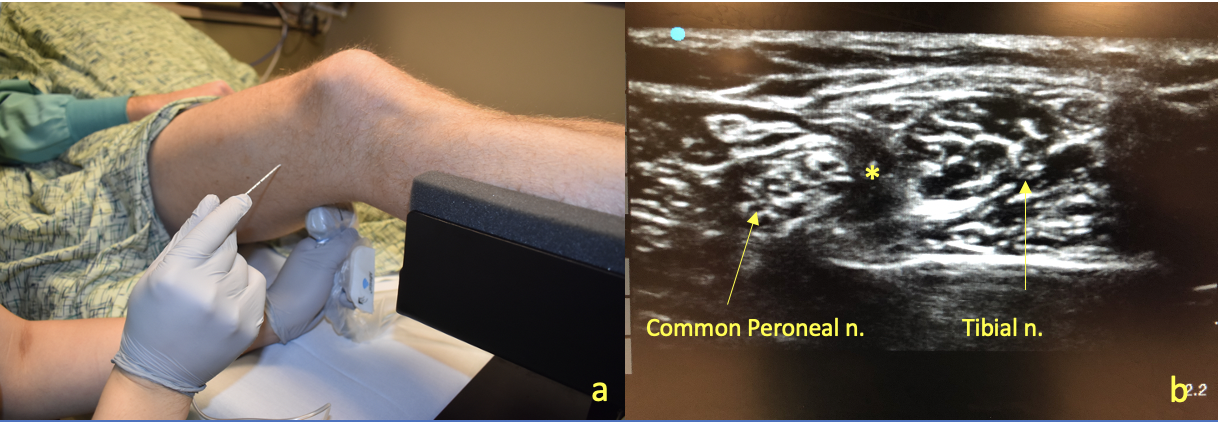
Figure 1a. Popliteal Approach to Sciatic Nerve Block. The leg is elevated on a foot pedestal to allow access of the ultrasound probe to scan the popliteal fossa. Figure 1b. Once the popliteal artery is identified, we scan proximally until the tibial nerve and common peroneal nerve joint together in the sciatic nerve sheath. Local anesthesia is injected subparaneurally (*).
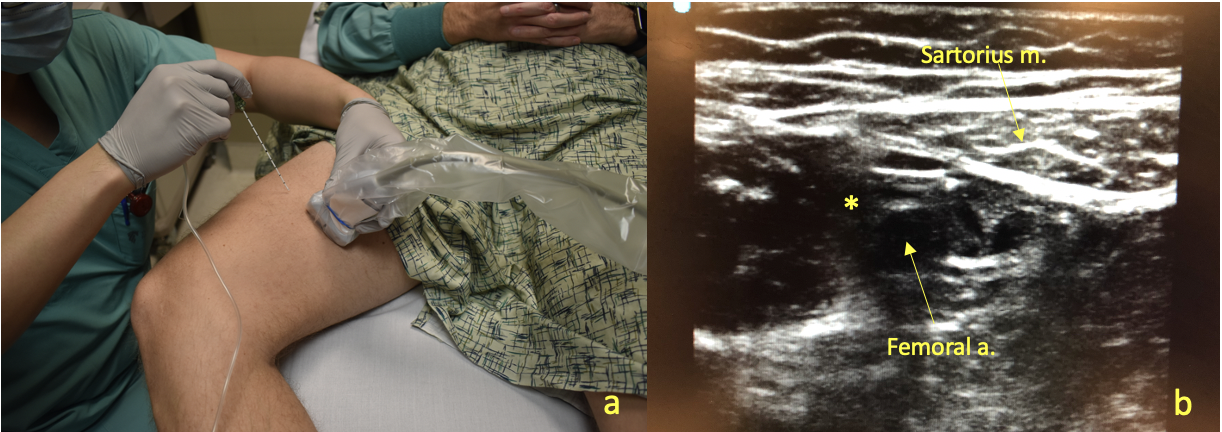
Figure 2a. Adductor Canal Block. The patient’s thigh is abducted and externally rotated to allow for adequate exposure of the medial aspect of the thigh. Figure 2b. Ultrasound imaging facilitates identification of the superficial femoral artery in the adductor canal posterior to the sartorius muscle. Needle insertion occurs antero-lateral and in-plane to the ultrasound probe until the needle tip is positioned deep to the sartorius muscle and anterior to the femoral artery, where local anesthetic (*) is deposited.
The popliteal sciatic nerve catheter will be placed first via the lateral approach; therefore, the patient will be placed in either the supine or lateral position. The surgical extremity is slightly flexed such that appropriate access to the popliteal crease is available. The patient’s skin should be cleansed with a disinfectant, and materials for the PNC should be sterilized to decrease the risk of infection. The ultrasound transducer should be placed in the transverse position beginning at the popliteal crease to facilitate identification of the pulsating popliteal artery. The ultrasound color doppler can assist in confirming the popliteal artery location and the tibial nerve is usually situated laterally and posteriorly to the popliteal vessels. Once the tibial nerve has been identified, the ultrasound probe is advanced proximally along the posterior leg until the tibial nerve can be visualized joining the common peroneal nerve to form the sciatic nerve. The bifurcation point should be around 5-10 cm from the popliteal crease. Catheter placement should occur at a point slightly proximal to the bifurcation of the sciatic nerve to ensure adequate blockade of both the tibial and common peroneal components. If difficulties are encountered with identification of sciatic nerve components, asking the patient to flex and extend their ankle may decrease ambiguity by demonstrating a “rocking horse” appearance of the tibial and peroneal nerves with ankle movement.
Once the ideal needle insertion site has been determined, local anesthetic should be injected to form a skin wheal. Under direct visualization, an 18G Tuohy needle should be advanced towards the sciatic nerve. Local anesthetic (20 mL of 0.25% bupivacaine with dexamethasone) is injected and circumferential spread around the targeted nerve structure can be seen. The nerve catheter can be threaded through the block needle and into the sciatic nerve sheath. The catheter should be advanced 4-5 cm beyond the needle tip and secured safely with adequate distance from the surgical site.
The saphenous nerve is anesthetized within the adductor canal. The patient should be placed in the supine position with the surgical lower limb externally rotated and flexed to allow for access to the medial thigh. After disinfecting the patient’s skin, the ultrasound probe can be applied in a sterile manner transversely between the middle and lower third of the thigh. The femoral artery must be identified first to find the saphenous nerve. The ultrasound laterally can be scanned laterally to identify the femur to establish the lateral border of the search for the femoral artery. Next, the pulsatile femoral artery can be identified with medial and cephalad scanning. Once the femoral artery is identified, the artery can be tracked distally until the sartorius muscle is located anterior to the femoral artery. The small saphenous nerve can be difficult to visualize under ultrasound. Local anesthetic should be deposited lateral to the femoral artery below the sartorius muscle. Incremental negative aspiration for blood to avoid a possible intravascular injection is performed prior to deposition of 10 mL of 0.25% bupivacaine with dexamethasone. The 20G multi-orifice PNC is inserted through the 18G Tuohy needle and secured safely in a location proximal to the surgical site. Communication with the surgical team about the proximity of the peripheral nerve catheter to the surgical site is crucial in order to avoid inadvertent dislodgement of the catheter. Other techniques to reduce premature catheter removal is the use of Dermabond to reduce leakage of local anesthetic around the catheter site and/or tunneling the nerve block catheter.
Regional Technique: Above Knee Amputation (AKA)
Providing adequate analgesia for AKA can be difficult due to the complex innervation of the upper leg and a lack of consensus regarding the optimal pain control regimen.8-10 The amputation site receives sensory innervation from multiple nerves, including the femoral, lateral femoral cutaneous, obturator, sciatic, and posterior femoral cutaneous nerves. The surgical field is also moved proximally to well above the knee, necessitating a different regional anesthetic plan than that commonly utilized for a BKA.
For all AKAs at our institution, our regional anesthetic plan consists of continuous femoral (Figure 3) and sciatic nerve blocks. Femoral nerve blocks are ultrasound-guided and performed in the supine position. Once the femoral nerve is identified underneath the fascia iliaca, local anesthetic (10 mL of 0.25% bupivacaine with dexamethasone) is injected, and a catheter is advanced to a perineural location.
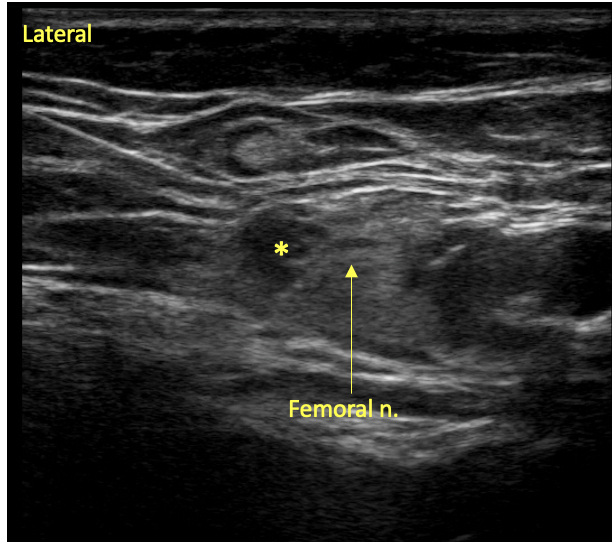
Figure 3. Femoral Nerve Block. The block needle is inserted from lateral to medial with spread of local anesthetic (*) visualized within the paraneural sheath.
The patient is then moved to the lateral position to undergo a transgluteal sciatic nerve block. This proximal approach to blockade of the sciatic nerve ensures that both the sciatic and posterior femoral cutaneous nerves are anesthetized. At our institution, the landmarks are usually identified via the Labat’s classical approach (Figure 4). After marking these landmarks, both an ultrasound and nerve stimulator are utilized to facilitate nerve localization and ensure success. Once the nerve is identified with ultrasound, the needle is approximated to the nerve and the nerve stimulator is adjusted to obtain an appropriate motor response (Figure 5). We attempt to obtain a motor response in the hamstring, calf, foot, and toes at a current threshold of 0.3-0.5 mA (possibly higher in the presence of neuropathy). Higher allowable current intensities may be acceptable and/or even desirable in the setting of diabetes or significant preexisting neuropathy. Following negative aspiration for blood, local anesthetic (20 mL of 0.25% bupivacaine with dexamethasone) is injected, and the needle may be adjusted to maximize the visualized local anesthetic spread pattern.
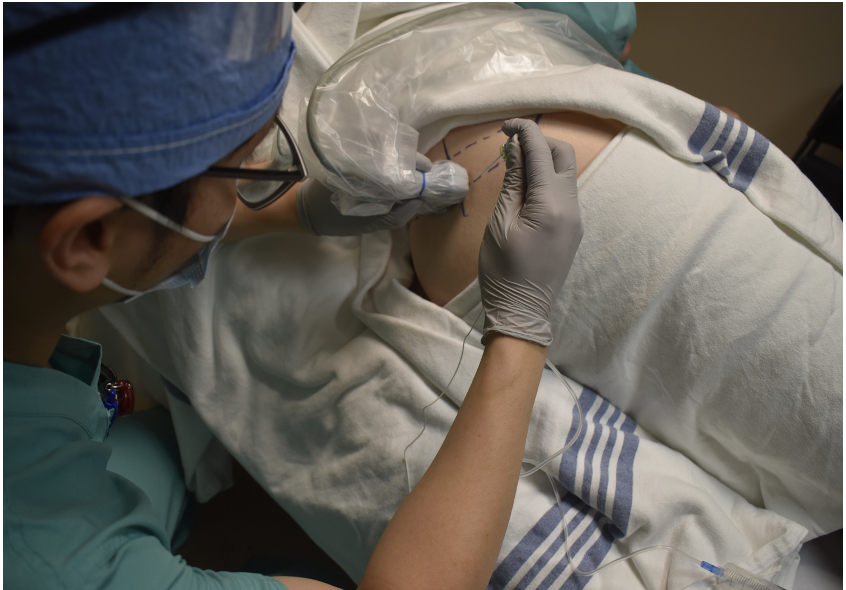
Figure 4. Labat’s Classical Approach. Patient in lateral position with the greater trochanter, posterior superior iliac spine, and sacral hiatus marked.
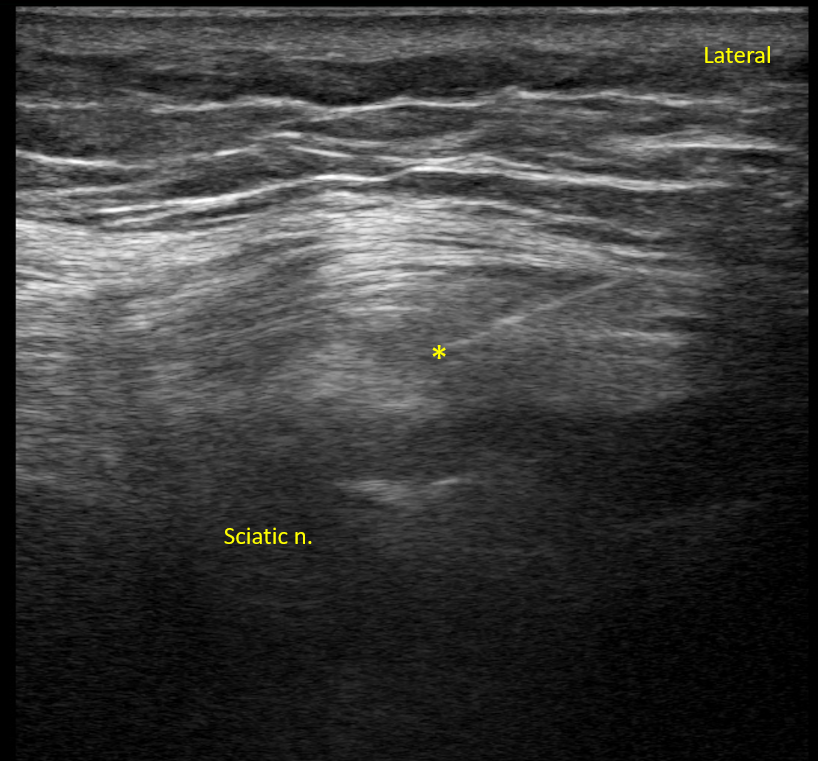
Figure 5. Sciatic Nerve Block. The needle is inserted from lateral to medial with spread of local anesthetic (*) visualized within the sciatic paraneural sheath.
Regional Technique: Anesthetic Plan
The intraoperative setting is a vital period where the primary anesthetic plan can vary with each patient. Comorbidities commonly present in this population may preclude general or neuraxial anesthesia. Regional anesthesia with sedation should be considered as the primary anesthetic in these scenarios.
One must consider neuraxial techniques when appropriate, as they also provide excellent analgesia for lower extremity amputations. Lambert et al. showed epidurals provided better post-amputation pain scores when compared to the nerve catheter group in the acute setting.11 However, the unique patient population that receives lower-limb amputation frequently has abnormal coagulation profiles or an ongoing anticoagulation treatment, which makes neuraxial techniques unfavorable due to an increased risk of epidural hematoma formation.
While epidurals and peripheral nerve blocks have been shown to provide excellent analgesia for post-amputation patients in the acute setting, no studies have demonstrated the ability of these procedures to prevent the development of chronic pain. Various studies have shown that PNC insertion following lower-limb amputation can reduce post-operative pain and opioid consumption while improving patient satisfaction. A systematic review demonstrated that PNC insertion reduced post-operative opioid consumption by about 50%.12 The current evidence suggests regional anesthesia helps optimize perioperative analgesia, but no firm conclusions can be drawn on whether this approach reduces the incidence or severity of CPAP.
Perioperative Multimodal Analgesia
In addition to regional anesthesia, it is important to continue non-opioid adjuvants such as gabapentin, acetaminophen, and NSAIDs, in an effort to minimize intraoperative opioid administration.
Ketamine, an NMDA receptor antagonist, is an effective analgesic that functions via blunting central pain sensitization.13 Intraoperative, subanesthetic ketamine infusions can help improve post-operative pain scores, and a ketamine infusion at a rate of 200 mcg/kg/hr is used in the intraoperative phase of care at our institution. Dexmedetomidine infusions can also be considered as a mechanism to inhibit nociceptive neurotransmission and provide a non-opioid adjuvant analgesia.14 In patients who have developed opioid tolerance or those with pain syndromes, dexmedetomidine infusions at a rate of 0.2-0.4mcg/kg/hr may be effectively used in the intraoperative phase of care.
Evidence for continuous PNCs decreasing pain and opioid consumption at 72 hours is inconclusive15; nonetheless, PNCs are continued 5-7 days post-placement to maximize potential analgesic benefits. The Acute Pain Service evaluates these patients each day that they are in the hospital and monitors for complications, such as infection or catheter migration. Depending on the post-placement day and site of insertion, we will discharge patients from the hospital with an elastomeric pump for continued analgesia. The Acute Pain Service contacts patients with elastomeric pumps in the home setting daily to document an evaluation of PNC efficacy of analgesia and absence of complications. This service has been utilized since 2014 without reported complications.
Conclusion
An aggressive multimodal analgesic regimen that incorporates PNCs is preferred at our institution. We believe this program strongly benefits our patients undergoing lower-extremity amputation. However, further study is needed to evaluate the long-term efficacy and perioperative timing of regional anesthesia and other analgesic adjuncts in reducing the risk of CPAP.
Duy A Do, MD, is a fellow in regional anesthesia and acute pain medicine in the department of anesthesiology at the University of Kansas Medical Center in Kansas City.

John-Paul J. Pozek, MD, is an assistant professor in the department of anesthesiology at the University of Kansas Medical Center in Kansas City.
References
- Johannesson A, Larsson G, Ramstrand N, Turkiewicz A, Wiréhn A, Atroshi I. Incidence of lower-limb amputation in the diabetic and nondiabetic general population: a 10-year population-based cohort study of initial unilateral and contralateral amputations and reamputations. Diabetes Care. 2008; 32(2):275-280. https://doi.org/10.2337/dc08-1639.
- Neil MJE. Pain after amputation. BJA Education. 2016; 16(3):107-112. https://doi.org/10.1093/bjaed/mkv028.
- Jensen TS, Krebs B, Nielsen J, Rasmussen P. Immediate and long-term phantom limb pain in amputees: Incidence, clinical characteristics and relationship to pre-amputation limb pain. Pain. 1985; 21:267–278. https://doi.org/10.1016/0304-3959(85)90090-9.
- Aladin H, Jennings A, Hodges M, Tameem A. Major lower limb amputation audit - introduction and implementation of a multimodal perioperative pain management guideline. Br J Pain. 2018; 12(4):230-237. https://doi.org/10.1177/2049463718769339.
- Karanikolas M, Aretha D, Tsolakis I, et al. Optimized perioperative analgesia reduces chronic phantom limb pain intensity, prevalence, and frequency: a prospective, randomized, clinical trial. Anesthesiology. 2011; 114(5):1144-1154. https://doi.org/10.1097/ALN.0b013e31820fc7d2.
- Mitra S, Sinatra RS. Perioperative management of acute pain in the opioid-dependent patient. Anesthesiology. 2004; 101(1):212-227. https://doi.org/10.1097/00000542-200407000-00032.
- Dirks J, Møiniche S, Hilsted K, Dahl JB. Mechanisms of postoperative pain: clinical indications for a contribution of central neuronal sensitization. Anesthesiology. 2002; 97(6):1591-1596. https://doi.org/10.1097/00000542-200212000-00035.
- Ayling OG, Montbriand J, Jiang J, et al. Continuous regional anaesthesia provides effective pain management and reduces opioid requirement following major lower limb amputation. Eur J Vasc Endovasc Surg. 2014; 48(5):559-564. https://doi.org/10.1016/j.ejvs.2014.07.002.
- De Jong R, Shysh AJ. Development of a multimodal analgesia protocol for perioperative acute pain management for lower limb amputation. Pain Res Manag. 2018; 2018:5237040. https://doi.org/10.1155/2018/5237040.
- Baddoo HK. A preliminary report on the use of peripheral nerve blocks for lower limb amputations. Ghana Med J. 2009; 43(1):24-28.
- Lambert AW, Dashfield AK, Cosgrove C, Wilkins DC, Walker AJ, Ashley S. Randomized prospective study comparing preoperative epidural and intraoperative perineural analgesia for the prevention of postoperative stump and phantom limb pain following major amputation. Reg Anesth Pain Med. 2001; 26(4):316-321. https://doi.org/10.1053/rapm.2001.23934.
- Bosanquet DC, Glasbey JC, Stimpson A, Williams IM, Twine CP. Systemic review and meta-analysis of the efficacy of perineural local anaesthetic catheters after major lower limb amputation. Eur J Vasc Surg. 2015; 50(2)241-249. https://doi.org/10.1016/j.ejvs.2015.04.030.
- Gorlin AW, Rosenfeld DM, Ramakrishna H. Intravenous sub-anesthetic ketamine for perioperative analgesia. J Anaesthesiol Clin Pharmacol. 2016; 32(2):160-167. https://doi.org/10.4103/0970-9185.182085.
- Tang C, Zhongyuan X. Dexmedetomidine in perioperative acute pain management: A non-opioid adjuvant analgesic. J Pain Res. 2017; 10:1899-1904. https://doi.org/10.2147/JPR.S139387.
- Ahuja V, Thapa D, Ghai B. Strategies for prevention of lower limb post-amputation pain: A clinical narrative review. J Anaesthesiol Clin Pharmacol. 2018; 34(4):439-449. https://doi.org/10.4103/joacp.JOACP_126_17 .
Leave a commentOrder by
Newest on top Oldest on top