POCUS Spotlight: Focused Cardiac Ultrasonography
Cite as: Sjaus A, Kalagara H. POCUS spotlight: focused cardiac ultrasonography. ASRA News. 2021;46. https://doi.org/10.52211/asra080121.047.
Introduction
There is little doubt that point-of-care ultrasound (POCUS) improves accuracy of assessment in perioperative care.1 Successful integration of POCUS into anesthesia practice requires psychomotor skills for image acquisition and cognitive skills for real-time interpretation and contextualization. Frequent hands-on practice and image reviews are the necessary commitments for those who wish to master these skills.2 Cardiac imaging can be challenging, and learners often benefit from expert guidance. In this article, we offer practical tips and tricks for using two-dimensional (2D) imaging to maximize image quality and utility of basic echocardiography.
A consistent, deliberate and systematic approach to basic aspects of FoCUS will maximize the clinical utility of cardiac ultrasound in the hands of novice sonographers. Anesthesiologists who wish to adopt the technique should scan often for variability and psychomotor skill development.
FOCUS Views and Terminology
Focused Cardiac Ultrasonography (FOCUS) or Focus Assessed Transthoracic Echo (FATE) views are standardized to specific tomographic planes selected to showcase the relevant cardiac structures. The five basic images are acquired through three echocardiographic windows (parasternal, apical, and subcostal). (Table 1) These areas on the thorax and abdomen allow unimpeded (by air or bone) transmission of ultrasound. There are many imaging planes/views for each window. The nomenclature names the window, then the plane/structure (“long axis”- aortic root in a longitudinal plane). Because FoCUS utilizes 2D echocardiography, understanding orthogonal imaging planes is important. (Figure 1)
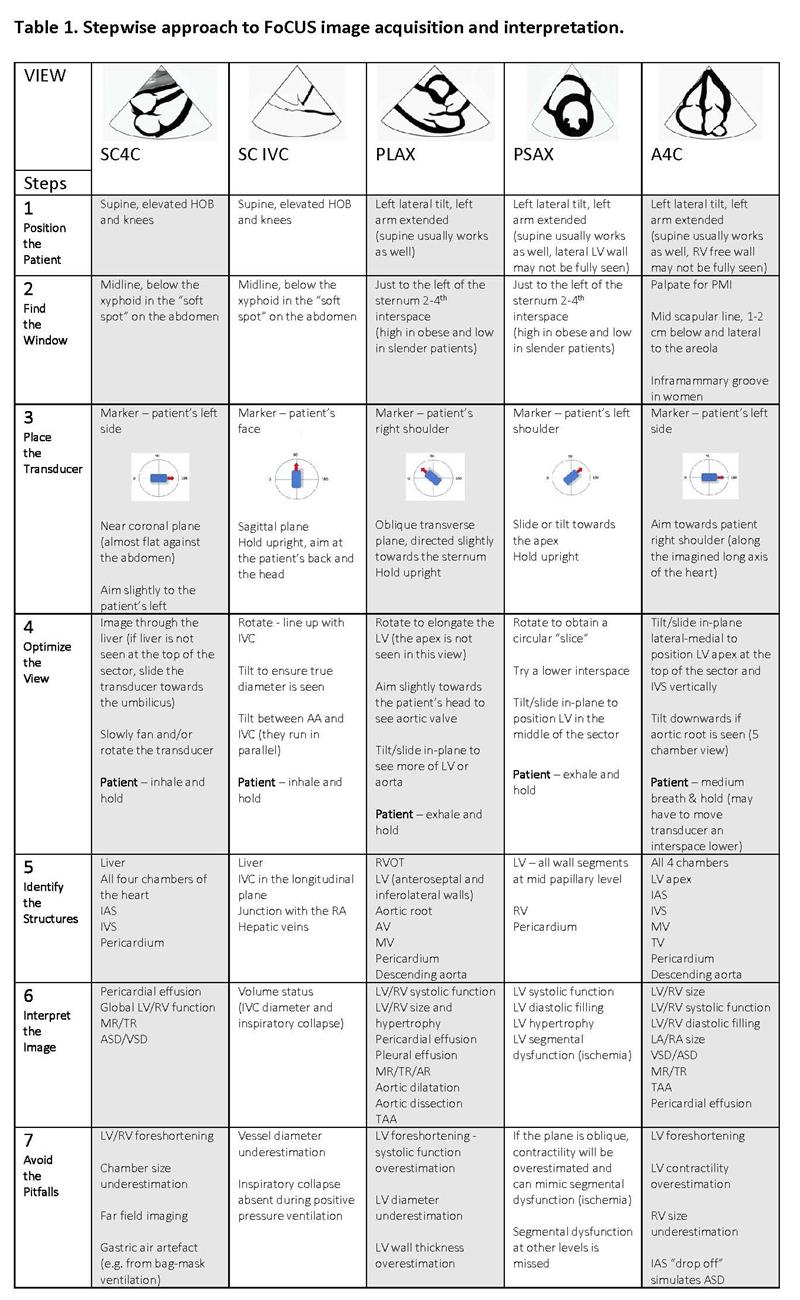
SC4C – subcostal four chamber view, SC IVC – subcostal view of the inferior vena cava, PLAX – parasternal long axis view, PSAX – parasternal short axis view, A4C – apical four chamber view, LV – left ventricle, RV – right ventricle, LA – left atrium, RA – right atrium, IAS – interatrial septum, IVS – interventricular septum, AV – aortic valve, MV – mitral valve, TV – tricuspid valve, IVC – inferior vena cava, MR – mitral regurgitation, TR – tricuspid regurgitation, AR – aortic regurgitation, TAA – thoracic aortic aneurysm, ASD – atrial septal defect, VSD ventricular septal defect
Imaging Sequence and Image Acquisition Tips
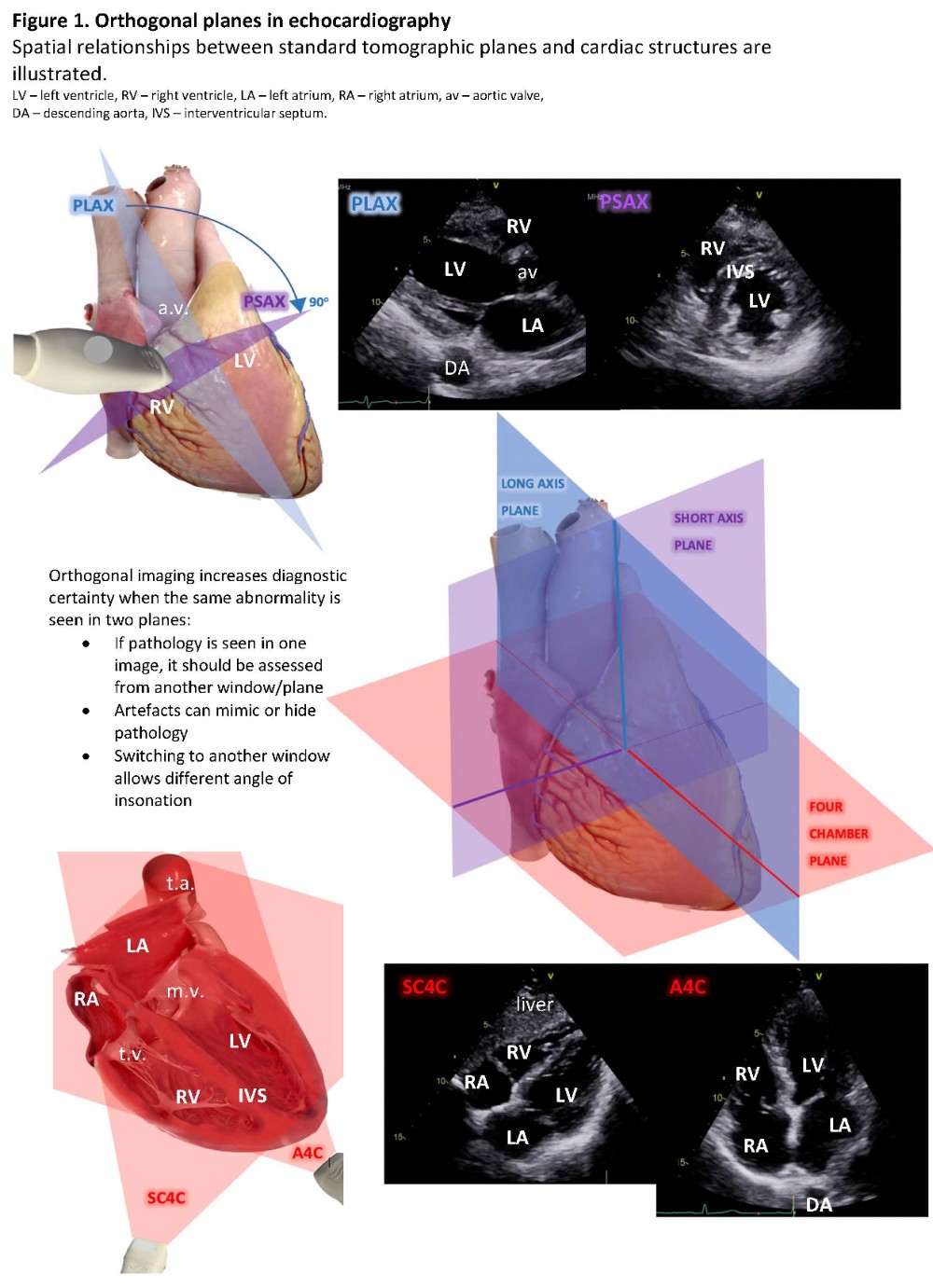
Regionalists are already experts in sonographic imaging. Phased array transducers have specific characteristics and optimizations. (Table 2) Adoption of a systematic approach that follows a sequential protocol ensures completeness.3
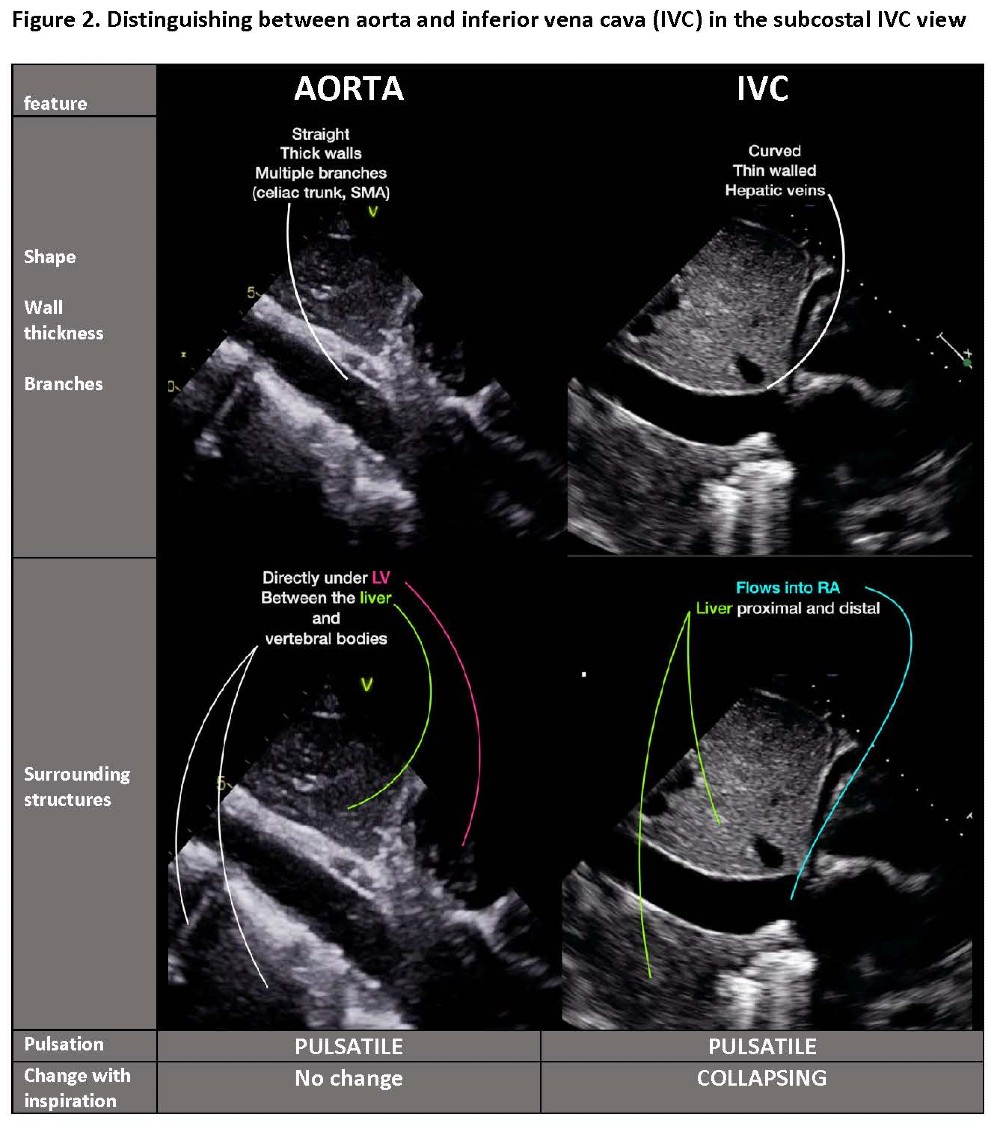
In the subcostal window, insonation is through the liver. The transducer is flat against the abdomen and directed slightly to the left to obtain the subcostal four chamber view (SC4C). In the same window, positioning sagittally midline with the marker pointing toward the patient’s head will show either abdominal aorta or the subcostal view of inferior vena cava (SCIVC). The vessels are parallel: tilting the transducer switches from aorta (left tilt) to IVC (right tilt). (Video 1, Figure 2)
Video 1. Anatomic relationship of abdominal aorta and inferior vena cava – easy imaging of both by tilting of transducer
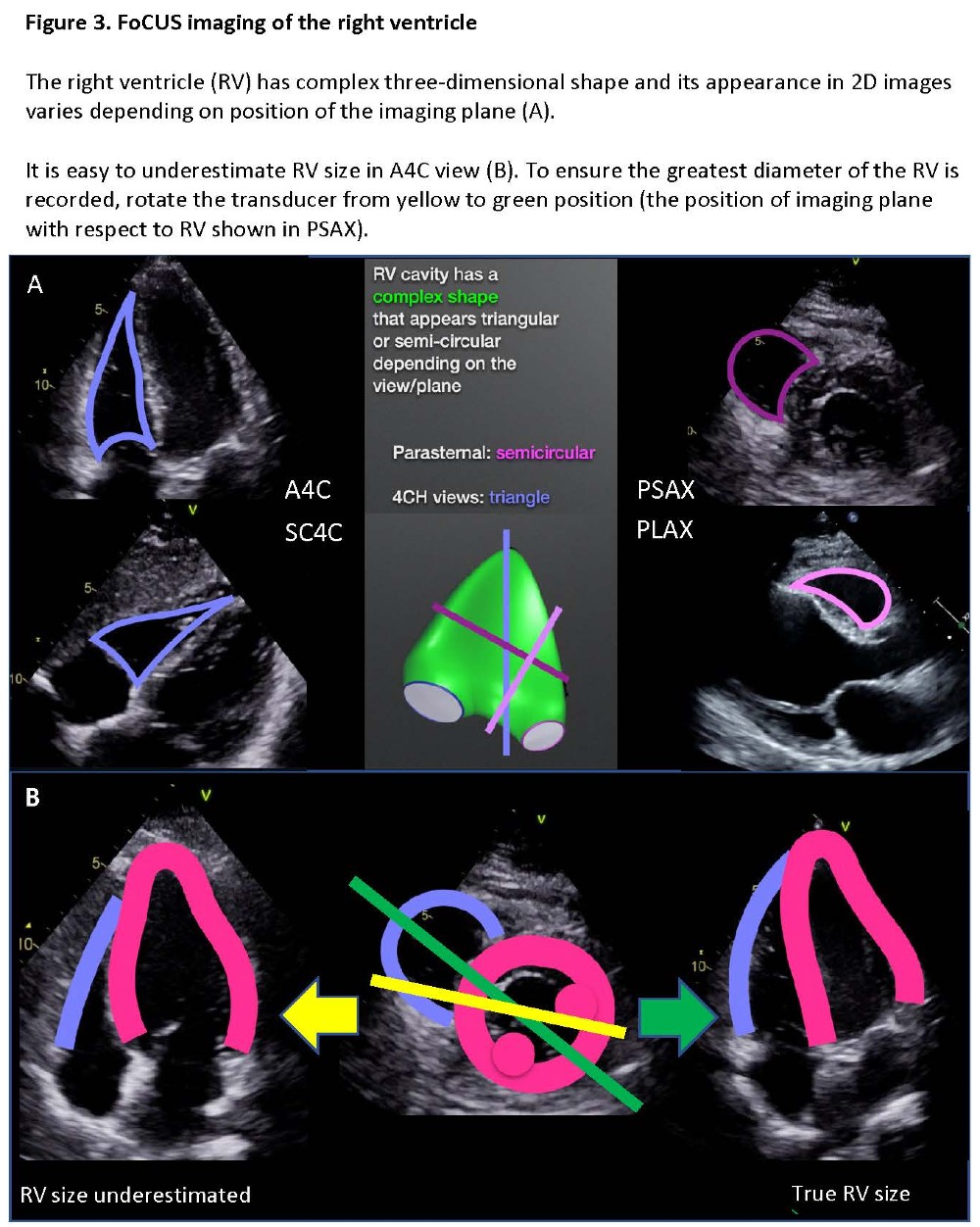
The apical window is located at the point of maximum impulse and is optimized with the patient tilted to the left. All chambers are seen in the four-chamber view (A4C) when the marker is to the patient’s left. Rotation of the transducer to visualize the largest right ventricular (RV) diameter is necessary to avoid underestimation of RV size. (Figure 3)
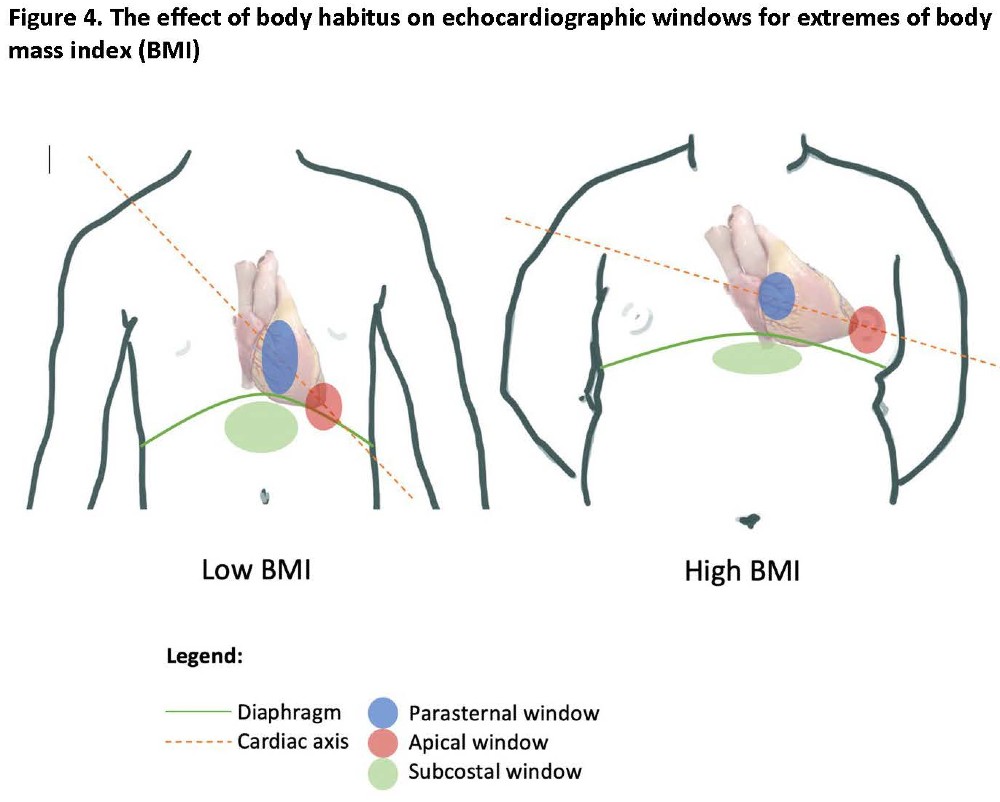
The parasternal window is easily accessible in supine patients. Its location along the sternal border is influenced by the patient’s body habitus. (Figure 4) The long axis view (PLAX) shows part of the right ventricle and all left-sided structures. From PLAX, the probe can be rotated 90o clockwise to obtain the short axis view (PSAX). By tilting the probe, the heart can be “sliced” mid ventricle (mid papillary), at apical or basal levels (through mitral or the aortic valves). For global left ventricular (LV) contractility and filling, use PSAX at the mid papillary level.
It is helpful to visualize the position of the heart within thorax: extending an imaginary line through the PLAX plane reveals the main axis of the heart. In pregnant or obese patients, this axis is more horizontal. (Figure 4)
Using a Patient’s Position and Breathing
Semi-left lateral tilt with the left forearm abducted is preferred. Back elevation can help in patients with a horizontal heart axis, while those with a vertical axis may require a head down position. Parasternal views are improved when the patient exhales. Conversely, inhalation of a mid-size breath may improve the A4C view by expanding the intercostal spaces and moving the heart anteriorly. For SC4C views, a deeper breath (avoiding Valsalva maneuver), head elevation and knee-up position will displace the heart toward the transducer and loosen the abdominal wall. In a mechanically ventilated patient, briefly pausing ventilation can improve visualization. Subcostal views may be the only ones accessible when lungs are hyperinflated. Gastric suctioning can improve subcostal images that are obscured by air.
Image Interpretation
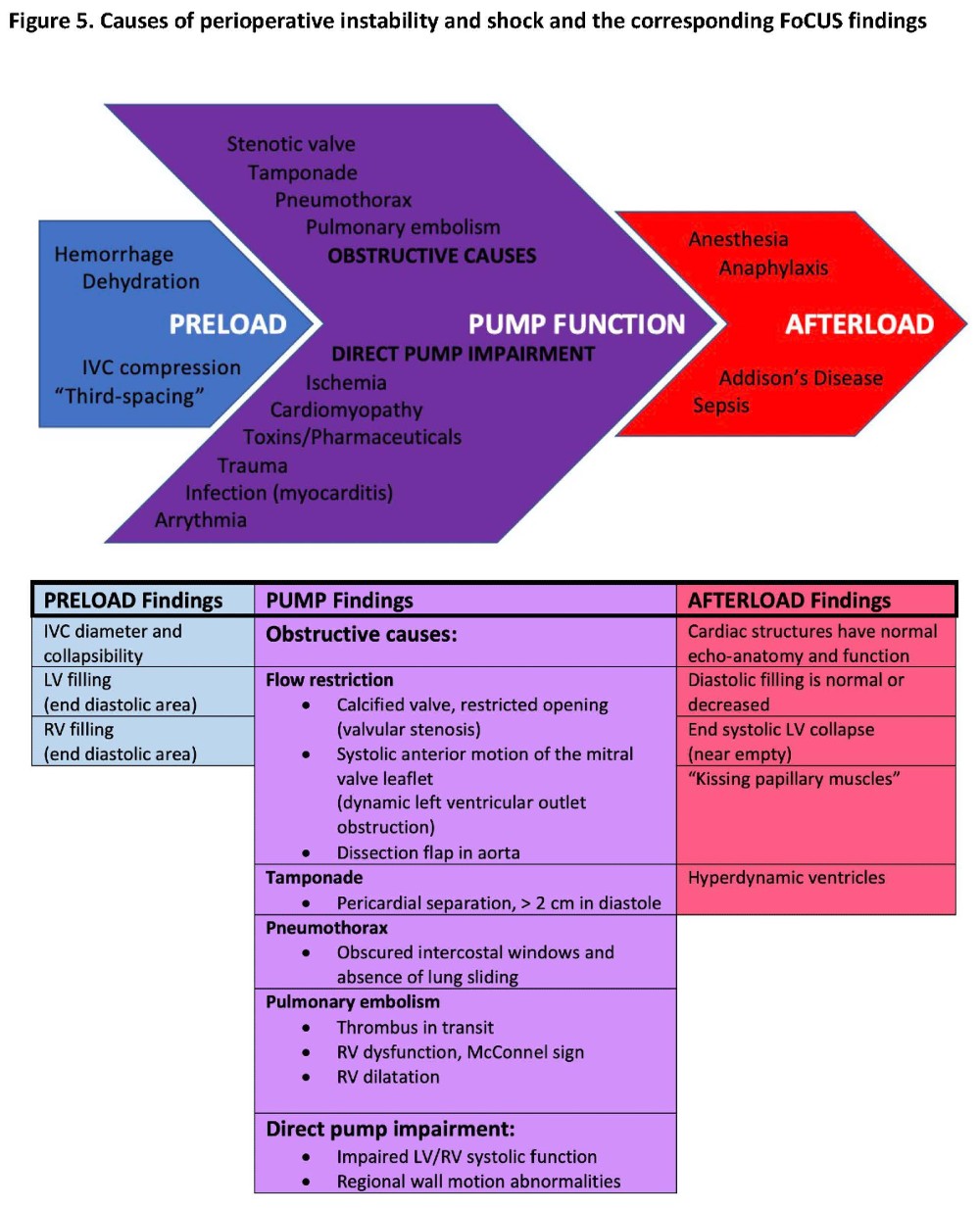
In most cases important clinical questions can be addressed through qualitative FoCUS assessment.4,5 (Figure 5) Viewing many loops of normal and diseased hearts will improve “calibration” of one’s eye. Tachycardia may result in an overestimation of contractility and underestimation of filling. Conversely, underestimation of systolic function may occur in bradycardia. (Video 2) Pitfalls include incorrect imaging planes (“foreshortening,” underestimating RV size) and artefacts. The added cognitive load of acquiring images at bedside and interpreting in real time increases the risk of errors.2 It is useful to pause a loop and advance frame by frame or “toggle” back and forth. If possible, it is important to save loops for a second look, especially when uncertain.
Video 2. Visual estimation of left ventricular contractility
Precise quantification during urgent bedside examination is considered unnecessary.6 However, semi-quantitative methods (fractional shortening and area change) can be useful.7 If required, size can be estimated from scale on the display.
1. Is the heart filled and contracting adequately?
Ventricular cavities expand and contract while the myocardium thins and thickens. The LV that fills well and completely empties in systole (hyperdynamic, “kissing” papillary muscles) is seen when systemic vascular resistance is low. Ventricles that appear small and empty completely, particularly when the IVC is narrow and collapses >50% on inspiration, suggest hypovolemia. A hypertrophic LV (diastolic wall thickness >1 cm) may give an impression of poor filling. This is the result of diastolic dysfunction rather than hypovolemia.
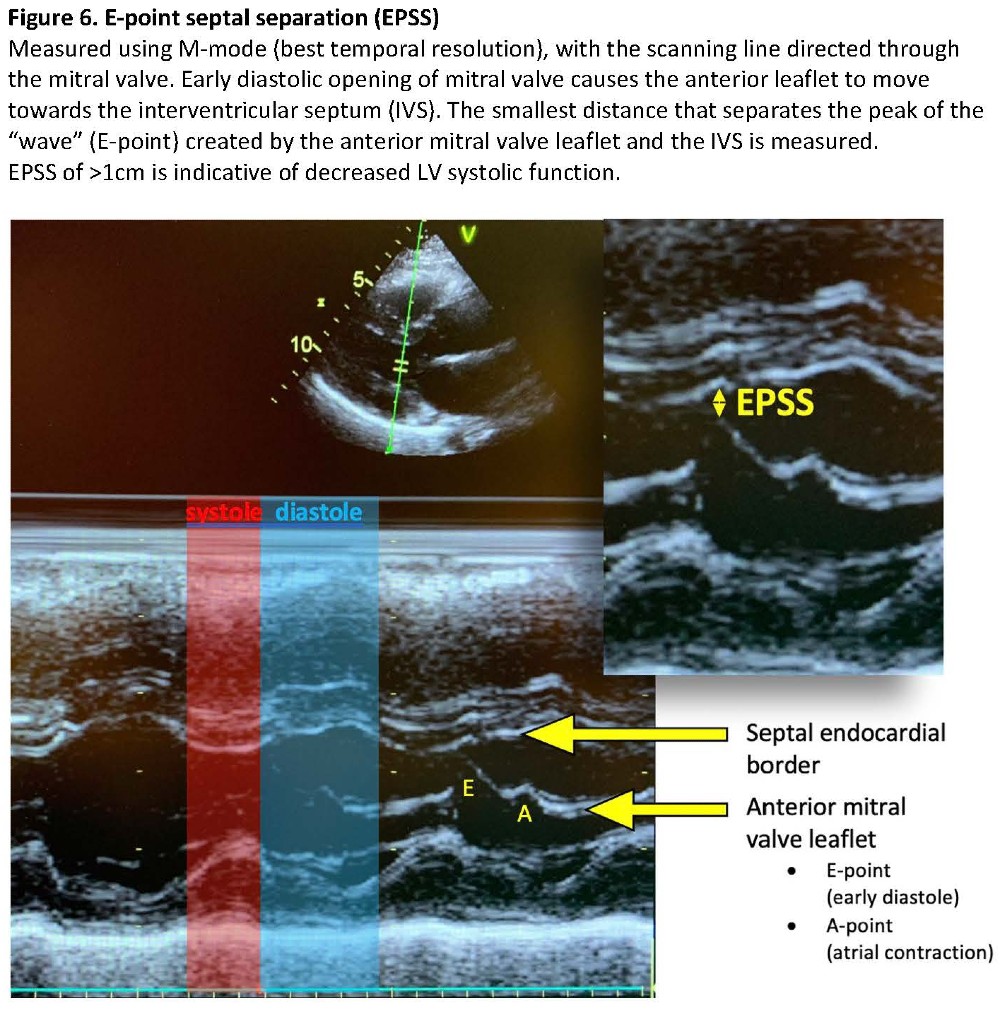
In systole, an LV contracting by less than a quarter of its diastolic diameter is functioning poorly. Mitral and aortic valves that have normal morphology but are opening incompletely with stroke volume support the diagnosis of systolic dysfunction. This is the basis of E-point septal separation (EPSS), a semiquantitative indicator of systolic function. (Figure 6) EPSS is a diastolic measurement performed using M-mode.8
A normal RV should have a smaller diameter when compared to the LV. Tricuspid annual plane excursion (TAPSE) contributes information about RV function. (Video 3)
Video 3. Tricuspid annular plane systolic excursion (TAPSE)
2. Can significant valvular abnormality be ruled out?
In five basic views, the valves are seen in longitudinal planes. Normal valves have thin, pliable mobile leaflets that open fully and coapt (close against each other) completely. Leaflets that do not close fully or prolapse may allow regurgitation. Calcified, thick, immobile leaflets may have stenosis or both. FoCUS does not quantify valvular lesions but, when combined with clinical assessment, can prompt changes in management and identify the need for advanced assessment. It is helpful to keep in mind that, in general, an abnormal-looking valve associated with normal chambers and vessels is less likely to present a hemodynamically significant lesion.
3. Can pericardial effusion/tamponade be ruled out?
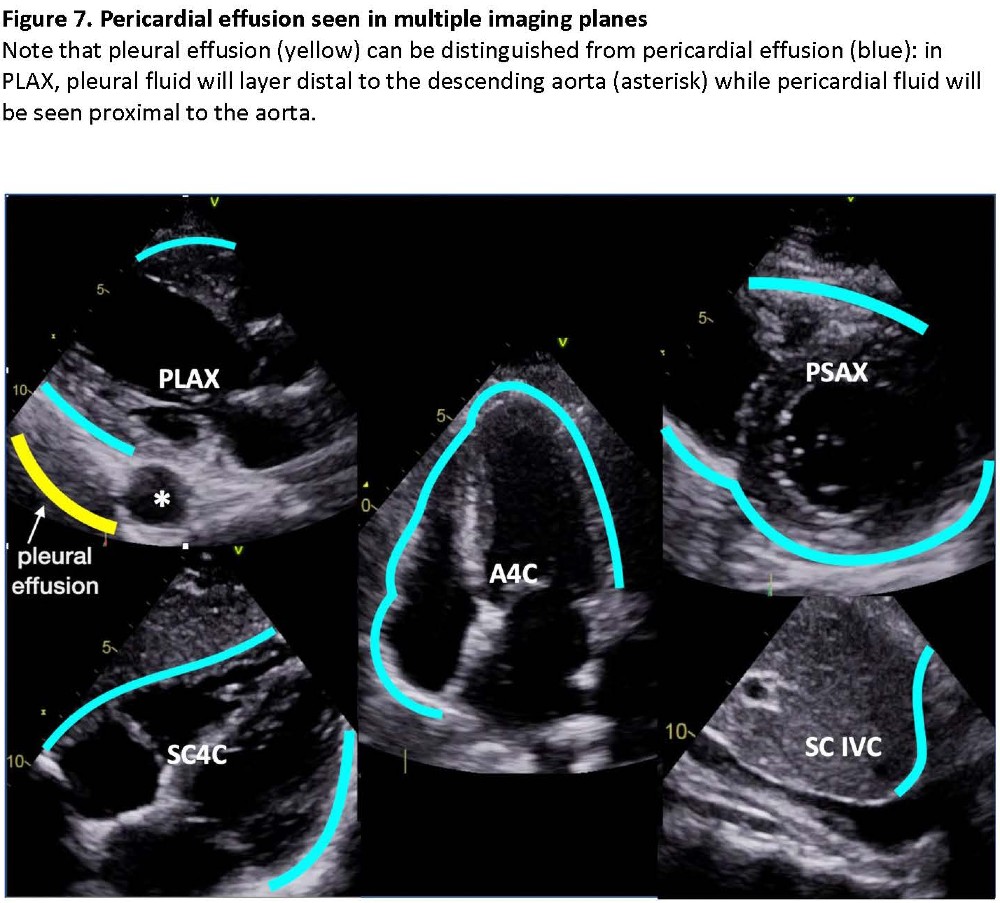
Pericardial effusion presents as an echo-lucent layer that changes width in systole and diastole. (Figure 7) In PLAX, pericardial effusions track between the heart and the descending aorta, while pleural effusions track behind the aorta – a distinguishing feature that can be easily missed if imaging depth is inadequate. Chronic effusions can be sizeable yet cause little hemodynamic compromise, while moderate acute effusions can cause diastolic collapse of the RV (tamponade). Hemopericardium, especially containing blood clots, can be difficult to image and requires advanced assessment.
4. Are there regional wall motion abnormalities (RWMA)?
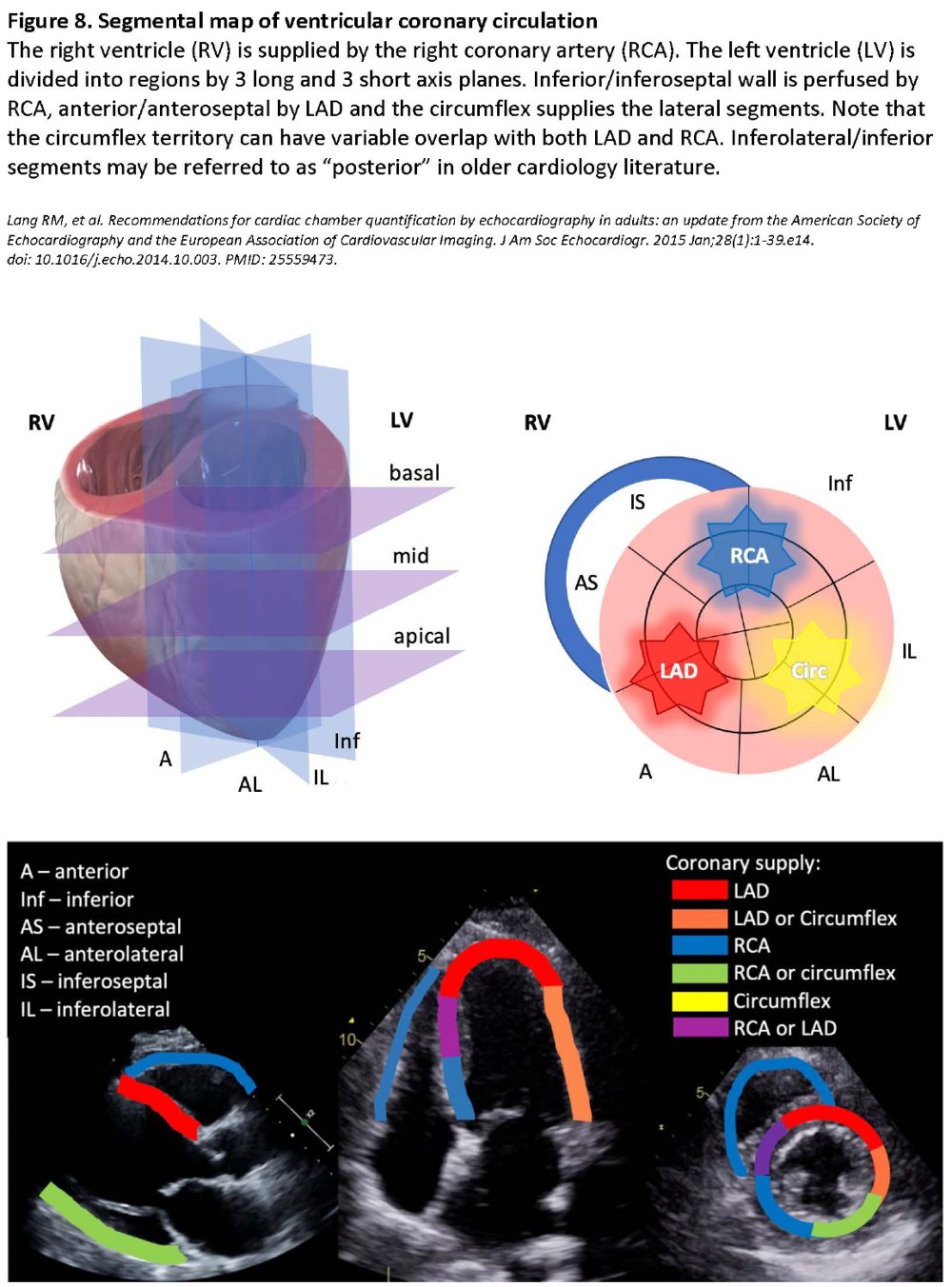
Myocardial dysfunction manifests as decreased systolic thickening and movement. Ischemia is implied if affected segments correspond to a coronary vessel territory. (Figure 8) These changes can be mimicked by a number of conditions and artefacts. Distinguishing new from old RWMAs can be challenging even for experienced echocardiographers.8 Therefore, RWMAs must be interpreted within clinical context.
Clinical Context
A defining feature of FoCUS is that it is intended as an adjunct to clinical assessment. The I-AIM approach (indication, acquisition, interpretation, management) can maximize the utility of FoCUS even when findings are unremarkable. Normal findings in an unstable patient will lead us down a different decision-making path than we would have chosen had potential cardiac causes remained uninvestigated. Hypovolemia is often clinically underestimated and is commonly discovered by FoCUS in volume-refractory perioperative hypotension.4 Finding poor systolic function, or a heavily calcified aortic valve, will similarly alter anesthetic management.
It is important to remember that divided attention during image acquisition can compete with vigilance and diligence in management. A prudent personal rule, especially in emergencies, is to deliberately limit scanning time and move on if the questions are not answered quickly.
Conclusion
A consistent, deliberate and systematic approach to basic aspects of FoCUS will maximize the clinical utility of cardiac ultrasound in the hands of novice sonographers. With an understanding of the basic principles stated in this article, anesthesiologists who wish to adopt the technique should scan often for variability and psychomotor skill development. In clinical practice, while we focus on performing FoCUS, we have to remember that imaging is the means – another item in our expert toolbox. For those who wish to build on the solid basic foundation, cardiac sonography can provide opportunities for progression toward more advanced skills. Even after years of fulfilling advanced echocardiography practice, we continue to find room to grow and new techniques to master.

Ana Sjaus, MD FRCPC, is an anesthesiologist in Women’s and Obstetric Anesthesia at IWK Health Centre and an assistant professor in the department of Anesthesiology, Pain Management and Perioperative Medicine at Dalhousie University in Halifax, Canada.

Hari Kalagara, MD, FCARCSI, EDRA, is an assistant professor in the department of Anesthesiology and Perioperative Medicine at Mayo Clinic in Jacksonville, FL.
References
- Ramsingh D, Runyon A, Gatling J, Dorotta, I Lauer R, Wailes D. et al. Improved diagnostic accuracy of pathology with the implementation of a perioperative point-of-care ultrasound service: quality improvement initiative. Reg Anesth Pain Med 2020;45:95-101. https://doi.org/10.1136/rapm-2019-100632.
- Thomas-Mohtat R, Sable C, Breslin K, Weinberg JG, Prasad A, Zinns L, et al. Interpretation errors in focused cardiac ultrasound by novice pediatric emergency medicine fellow sonologists. Crit Ultrasound J 2018;J10:33. https://doi.org/10.1186/s13089-018-0113-4.
- Nagre AS. Focus-assessed transthoracic echocardiography: Implications in perioperative and intensive care. Ann Card Anaesth. 2019;22(3):302-8. https://doi.org/10.4103/aca.ACA-88-18.
- Markin NW, Gmelch BS, Griffee MJ, et al. A Review of 364 perioperative rescue echocardiograms: findings of an anesthesiologist-staffed perioperative echocardiography service. J Cardiothorac Vac Anesth 2015;29(1): 82-8. https://doi.org/10.1053/j.jvca.2014.07.004.
- Kratz T, Holtz S, Thorsten S, Steinfeldt T, Exner M, Campo dell’Orto M, et al. Feasibility and impact of focused intraoperative transthoracic echocardiography on management in thoracic surgery patients: an observational study. J Cardiothorac Vac Anesth 2018;32(2):848-52. https://doi.org/10.1053/j.jvca.2017.09.010.
- Spencer KT, Kimura BJ, Korcarz CE, Pellikka PA, Rahko PS, Siegel RJ. Focused cardiac ultrasound: recommendations from the American Society of Echocardiography. J Am Soc Echocardiogr 2013;26(6):567-81. https://doi.org/10.1016/j.echo.2013.04.001.
- McKaigney CJ, Krantz MJ, La Rocque CL, Hurst ND, Buchanan MS, Kendall JL. E-point septal separation: a bedside tool for emergency physician assessment of left ventricular ejection fraction. Am J Emerg Med 2014;32(6):493-7. https://doi.org/10.1016/j.ajem.2014.01.045.
- Lang RM, Badano LP, Mor-Avi V, Afilalo J, Armstrong A, Ernande L, et al. Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr 2015;28(1):1-39.e14. https://doi.org/10.1016/j.echo.2014.10.003.
- Johnson B, Lovallo E, Frenkel O, Nagdev A. Detect Cardiac Regional Wall Motion Abnormalities by Point-of-Care Echocardiography. ACEP Now. https://www.acepnow.com/article/detect-cardiac-regional-wall-motion-abnormalities-point-care-echocardiography/?singlepage=1&theme=print-friendly. Accessed March 2021.
Leave a commentOrder by
Newest on top Oldest on top