Cervicogenic Headache
Author
Samer Narouze, MD, PhD
Clinical Professor of Anesthesiology and Pain Management, Ohio University
Clinical Professor of Neurological Surgery, Ohio State University
Chairman, Center for Pain Medicine
Western Reserve Hospital
Cuyahoga Falls, OH
Introduction
Cervicogenic headache was initially defined as unilateral headache that is provoked by neck movement or pressure over tender points in the neck with associated reduced range of movement of the cervical spine. The headache occurs in non-clustering episodes and is usually non-throbbing in nature, originating from the neck and spreading over the occipital, temporal, and frontal regions.[1-3]
However, these clinical criteria are not enough to make a definite diagnosis of cervicogenic headache, as it is sometimes difficult to differentiate clinically between cervicogenic headache, migraine, and tension-type headache.[4-6]
Response to diagnostic block of the nerve supply of these cervical structures or intraarticular injection of local anesthetic into the culprit joint is now considered the major crtierion in the diganosis of cervicogenic headache.[7]
Also cervicogenic headache can be unilateral or bilateral.[7]
These clinical findings prompted the development of new diagnostic criteria for the diagnosis of cervicogenic headache by the International Headache Society (IHS) in 2004 (ICHD-2nd edition). More recently, the ICHD-3rd edition was released.[8]
Diagnostic Criteria
A. Any headache fulfilling criterion C
B. Clinical, laboratory and/or imaging evidence of a disorder or lesion within the cervical spine or soft tissues of the neck, known to be able to cause headache.
C. Evidence of causation demonstrated by at least two of the following:
- Headache has developed in temporal relation to the onset of the cervical disorder or appearance of the lesion.
- Headache has significantly improved or resolved in parallel with improvement in or resolution of the cervical disorder or lesion.
- Cervical range of motion is reduced and headache is made significantly worse by provocative maneuvers.
- Headache is abolished following diagnostic blockade of a cervical structure or its nerve supply
D. Not better accounted for by another ICHD-3 diagnosis.
Etiology
Cervicogenic headache is referred pain from cervical structures innervated by the upper three cervical spinal nerves. Thus possible sources of cervicogenic headache are: atlanto-occipital joint, median and lateral atlanto-axial joints, C2-3 intervertebral disc, C2-3 zygapophysial joint, upper posterior neck and paravertebral muscles, the trapezius and the sternocleidomastoid muscles, spinal and posterior cranial fossa dura matter, cervical spinal nerves and roots, and the vertebral artery.[9]
Neuroanatomy and Neurophysiology
The spinal nucleus of the trigeminal nerve extends caudally to the outer lamina of the dorsal horn of the upper three to four cervical spinal segments. This is known as the trigeminocervical nucleus, which receives afferents from the trigeminal nerve as well as the upper three cervical spinal nerves.
Convergence between these afferents accounts for the cervical-trigeminal pain referral. Therefore, pain originating from cervical structures supplied by the upper cervical spinal nerves could be perceived in areas innervated by the trigeminal nerves such as the orbit and the frontotemporoparietal region.
Bartsch and Goadsby showed that noxious stimulation of the greater occipital nerve induces increased central excitability of supratentorial afferents[10] and vice versa. Stimulation of the dura mater increases trigeminocervical neurons responsiveness to cervical input.[11]
Common Sources of Cervicogenic Headache
We will discuss here the clinical presentations of the common causes of cervicogenic headache and how to come up with an accurate diagnosis and plan of care[12] (Figure 1).
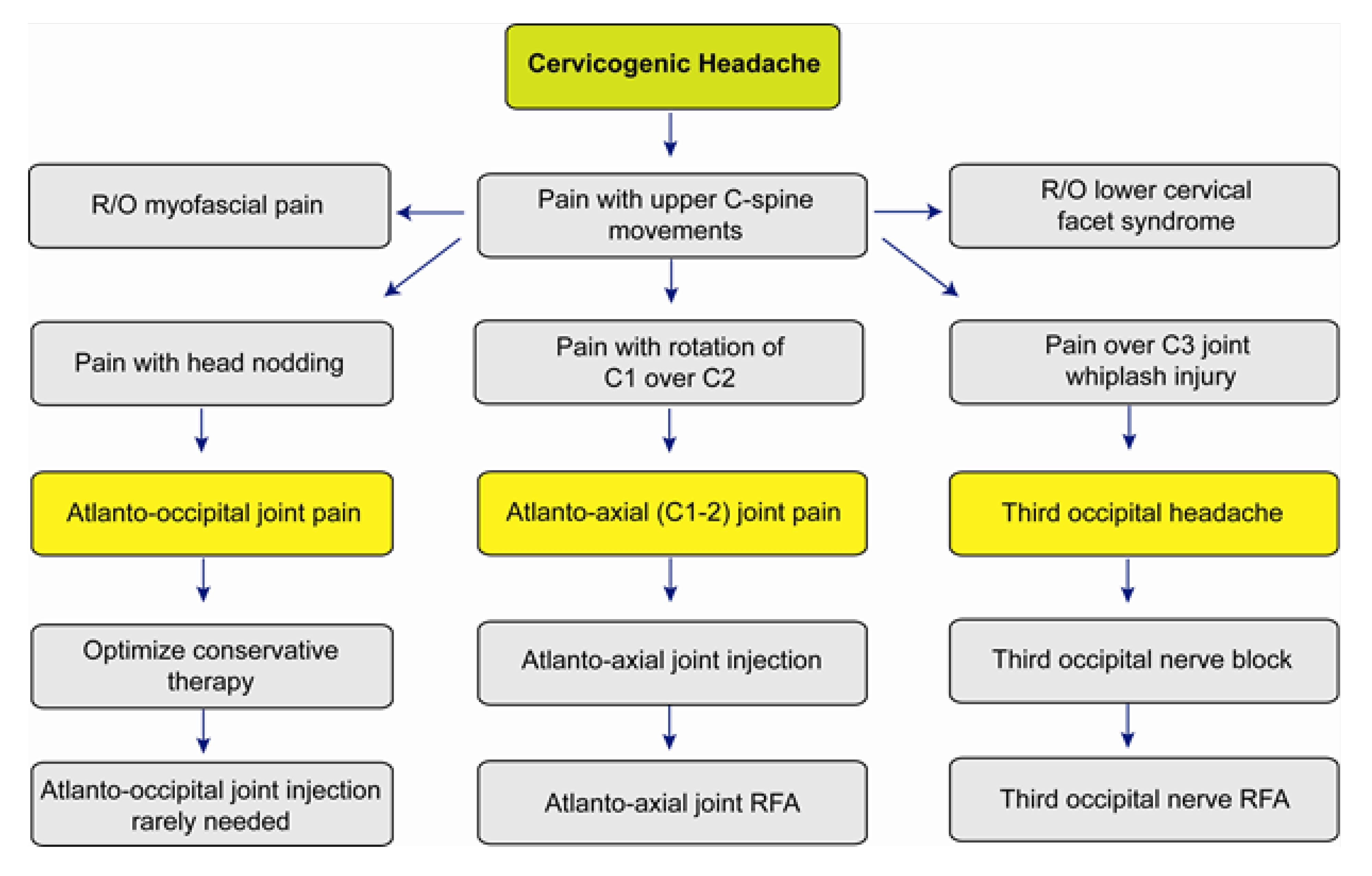
Figure 1: Algorithm for the diagnosis and management of cervicogenic headache. (Reprinted with permission from Narouze S, ed. Interventional management of head and face pain: Nerve blocks and beyond.
Atlanto-Axial joint
The lateral atlanto-axial joint, which is innervated by the C2 ventral ramus, is not an uncommon cause of cervicogenic headache. It may account for 16% of patients with occipital headache.[13] In human volunteers distending the lateral atlanto-axial joint with contrast agent produces occipital pain.[13]
Clinical presentations suggestive of pain originating from the lateral atlanto-axial joint include: occipital or suboccipital pain, focal tenderness over the suboccipital area or over the transverse process of C1, restricted painful rotation of C1 on C2, and pain provocation by passive rotation of C1.
These clinical presentations merely indicate that the lateral atlanto-axial joint could be a possible source of occipital headache, however they are not specific and therefore cannot be used alone to establish the diagnosis.[9]
The only means of establishing a definite diagnosis is a diagnostic block with intra-articular injection of local anesthetic.[12] The pathology of lateral atlantoaxial joint pain is usually osteroarthritis or post-traumatic in nature.[14,15] However, the presence of osteroarthritic changes in imaging studies doesn't mean that the joint is necessary painful; also, the absence of abnormal findings doesn't preclude the joint from being painful.
There is no conservative treatment for lateral atlanto-axial joint pain. However intra-articular steroids are effective in short-term relief of pain originating from the lateral atlanto-axial joint.[16-17] Long-lasting pain relief may require arthrodesis of the lateral atlanto-axial joint.[18-19]
Atlanto-axial joint intra-articular injection has the potential for serious complications, so it is imperative to be familiar with the anatomy of the joint in relation to the surrounding vascular and neural structures (Figure 2).
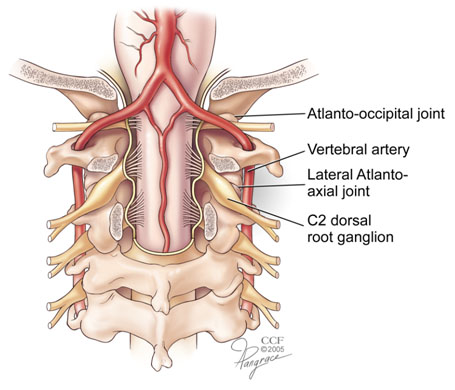
Figure 2. Illustration showing the relationship of the atlantoaxial and atlantoccipital joints to the vertebral artery.
The vertebral artery lies lateral to the atlanto-axial joint as it courses through the C2 and C1 foramina. Then it curves medially to go through the foramen magnum crossing the medial posterior aspect of the atlanto-occipital joint (Figure 2).
The C2 dorsal root ganglion and nerve root with its surrounding dural sleeve crosses the posterior aspect of the middle of the joint. Therefore, during atlanto-axial joint injection, the needle should be directed toward the posterolateral aspect of the joint. This will avoid injury to the C2 nerve root medially or the vertebral artery laterally (Figures 3-5).[12][17]
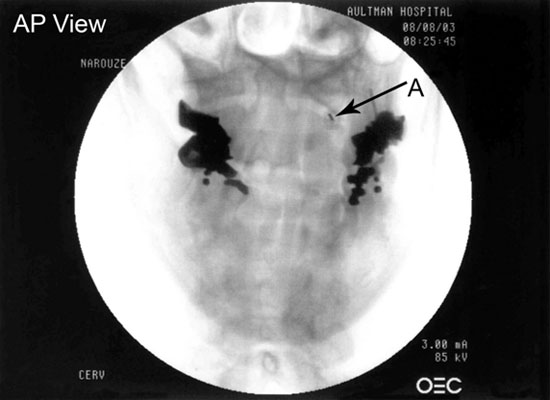
Figure 3. AP view showing the needle in a tunnel view inside the lateral part of the lateral AA joint.
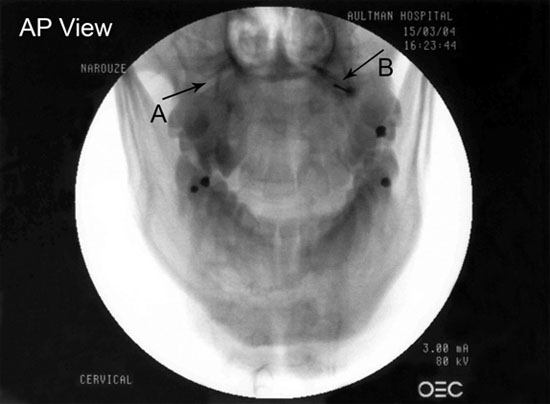
Figure 4. AP view showing the lateral atlanto-axial joint (A) and the needle tip and contrast agent within the lateral AA joint (B).
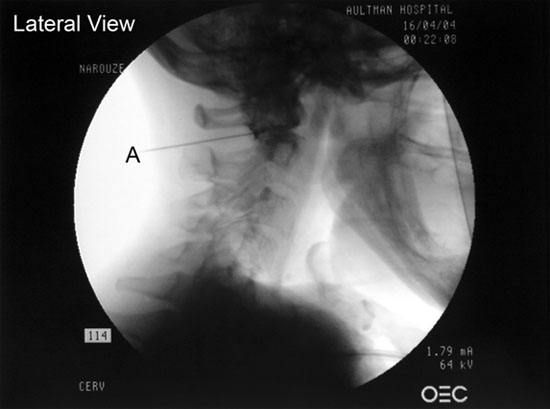
Figure 5. Lateral view showing the needle tip and contrast agent within the lateral AA joint.
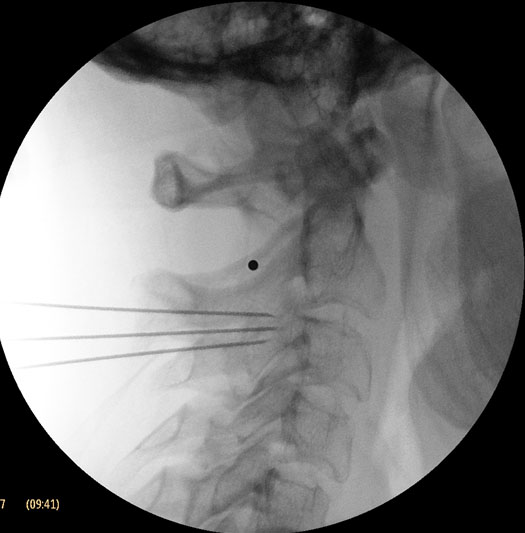
Figure 6. Lateral view showing three radiofrequency needles appropriately placed, at the equator of the C2-3 joint, above and below the joint line.
Meticulous attention should be paid to avoid intravascular injection as the anatomy may be variable. Injection of a contrast agent should be performed under real time fluoroscopy preferably with digital subtraction prior to injection of the local anesthetic as negative aspiration is not reliable.
Inadvertent puncture of the C2 dural sleeve with CSF leak or high spinal spread of the local anesthetic may occur with atlanto-axial joint injection if the needle is directed only a few millimeters medially.
Spinal cord injury and syringomyelia are potential serious complications if the needle is directed further medially.
C2-3 Zygaphphysial Joint and Third Occipital Nerve
The C2-3 zygapophyseal joint is innervated by the third occipital nerve, which is the superficial medial branch of the dorsal ramus of C3.[20]
Pain stemming from this joint (named third occipital headache) was seen in 27% of patients presenting with cervicogenic headache after a whiplash injury.[21]
Tenderness over the C2-3 joint is only suggestive of third occipital headache, a controlled diagnostic third occipital nerve block is mandatory to confirm the diagnosis. Earlier reports showed that radiofrequency neurotomy of the third occipital nerve were not effective[22], however with improved radiofrequency technique; complete relief was obtained in 88% of patients with third occipital headache (Figure 6).[23]
C2-3 zygapophyseal joint intra-articular steroid injection was effective in one study.[24] On the other hand, Barnsley et al. reported a lack of efficacy of intra-articular steroids for chronic pain stemming from the cervical zygapophyseal joints.[25]
Third occipital nerve neurolysis
The third occipital nerve is the superficial medial branch of C3 dorsal ramus. It supplies the C2-3 zygapophysial joint while crossing the joint laterally. Also it supplies part of the semispinalis capitis muscle and its cutaneous branch supplies a small area of skin below the occiput.[20]
Third occipital radiofrequency neurolysis was shown to be effective in the treatment of headache stemming from the C2-3 joint. The most common side effect is incomplete lesioning of the third occipital nerve because of its variable anatomy. [22]
The use of the three needles technique to accommodate all variation in the anatomy of the third occipital nerve from just lateral to the joint line to above or below the joint, and creating consecutive lesions no more than one electrode width from adjacent lesion markedly improved the results (Figure 6).[23]
Anesthesia dolorosa has not been reported with this particular block. However, numbness in the cutaneous distribution of the nerve is very common, whereas dysesthesia and hypersensitivity typically at the border of the area of numbness occur in up to 50% of cases. These are temporary complications that usually persists only for a few days to weeks.[22-23]
Temporary ataxia has been reported in most patients as third occipital neurotomy partially denervates the semispinalis capitis muscles with the resultant interference of the tonic neck reflexes. Most patients can overcome this sensation by relying on visual cues.[22-23]
Other possible complications which are not specific to third occipital nerve block, as they may occur with cervical medial branch blocks at lower levels include: infection, hematoma formation, and injury to the vertebral artery or the cervical nerve root if the needle is not positioned appropriately.
Occipital Neuralgia Nerve
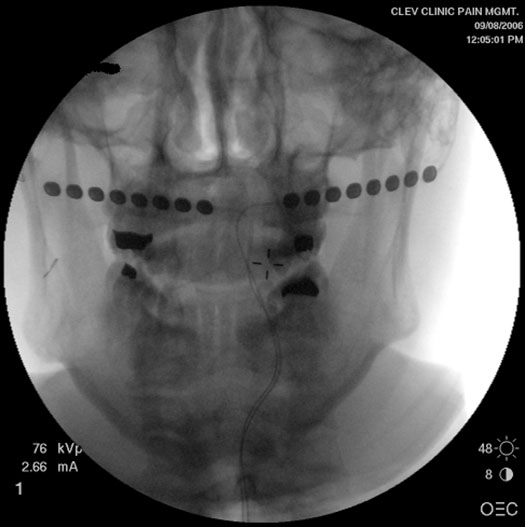 Figure 7. AP view showing bilateral occipital surgical leads.
Figure 7. AP view showing bilateral occipital surgical leads.
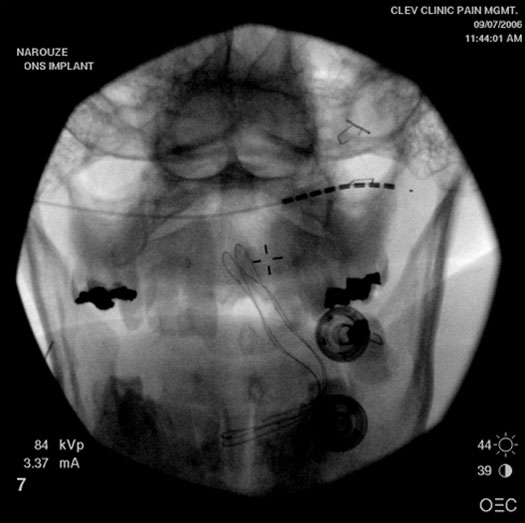 Figure 8. AP view showing right occipital percutaneous lead.
Figure 8. AP view showing right occipital percutaneous lead.
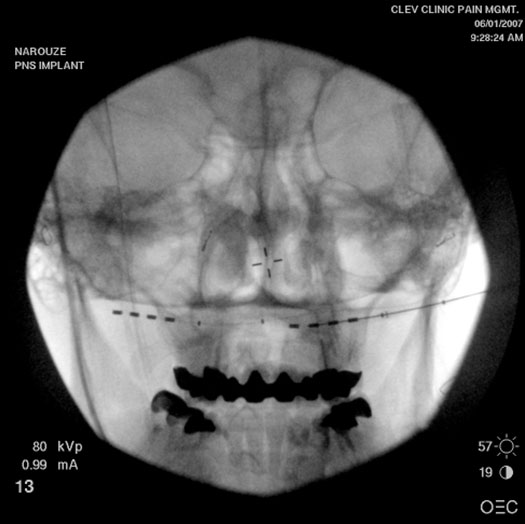 Figure 9. AP view showing bilateral occipital self anchoring leads.
Figure 9. AP view showing bilateral occipital self anchoring leads.
According to the International Classification of Headache Disorders (ICHD), occipital neuralgia is coded separately under cranial neuralgias.[8] It is discussed because of its relevance to cervicogenic headaches.
A. Paroxysmal stabbing pain, with or without persistent aching between paroxysms, in the distribution(s) of the greater, lesser and/or third occipital nerves
B. Tenderness over the affected nerve
C. Pain is eased temporarily by local anesthetic block of the nerve
Occipital neuralgia was long through to be the result of entrapment of the greater occipital nerve as it emerges from the trapezius muscle. However, surgical nerve release gives only short-term relief in about 80% of cases, while nerve excision provides short-term relief in about 70% of patients.[26-27]
Occipital neuralgia must be distinguished from occipital referral of pain from the atlantoaxial or upper zygapophyseal joints or from tender trigger points in neck muscles or their insertions.[8] The greater occipital nerve is the terminal branch of the dorsal ramus of C2 with contribution from C3 while the lesser occipital nerve is a branch of the dorsal ramus of C3 with contributions from C2. Segmental nerve blocks at C2 and C3 may be necessary to make the diagnosis in some cases.[28]
Cryoneurolysis, radiofrequency ablation, and more permanent neuroablative approaches as dorsal rhizotomy at C1-3 and partial posterior rhizotomy at C1-3 showed variable responses.[29-32] Recently; there has been a growing interest in pulsed radiofrequency (PRF) for the treatment of intractable occipital neuralgia. To date; there are 2 prospective studies and one retrospective multicenter study with promising results.[33-35] However; the lack of randomized controlled studies and the variation in the technique with landmark versus imaging guidance; limit the wide application of this treatment modality.
Occipital Neurostimulation
Percutaneous occipital nerve stimulation, unlike neuroablative techniques, offer the potential for a minimally invasive, low risk, and reversible approach to managing occipital neuralgia, chronic migraine, cluster headaches, and other types of intractable primary headaches.[36-38]
PET scan studies showed increased regional cerebral blood flow in areas involved in central neuromodulation in chronic migraine patients treated with occipital nerve electrical stimulation.[39] A percutaneous trial of peripheral nerve stimulation is performed using subcutaneous electrodes placed superficial to the cervical muscular fascia at the level of the C1 or the nuchal line. If effective, a permanent implant may be carried out using the same electrode lead type or paddle-type surgical lead and attached to a pulse generator implanted in the infraclavicular area, abdomen, flank, or upper buttock (Figures 6 and 7).
The ONSTIM (occipital nerve stimulation for the treatment of intractable migraine) trial, examined the safety and efficacy of ONS.[40] It is a multicenter prospective randomized single-blind controlled feasibility study. Patients who responded favorably to occipital nerve block (ONB) were randomized (2:1:1) into 3 groups: adjustable stimulation (AS), preset stimulation (PS), or medical management (MM). Those who did not respond to ONB formed an ancillary group (AG). One hundred and ten patients were enrolled from 9 centers. Sixty sex subjects completed diary data during 3-month follow-up. At 3 months, percent reduction in headache days/month was 27.0% (AS), 8.8% (PS) (p = 0.132), 4.4% (MM) (p = 0.058), 39.9% (AG) (p = 0.566). Responder rate was 39% (AS), 6% (PS) (p =0.032), 0% (MM) (p = 0.003), and 40% (AG) (p = 1.000).
More recently, another prospective, multi-center, double-blind, controlled study of ONS in patients with CM.[41] One hundred and fifty-seven patients from 15 centers were enrolled and randomized (2:1) to either a stimulation trial followed by device implantation and active stimulation for 12 weeks (n = 105) or a stimulation trial followed by device implantation but sham stimulation for 12 weeks (n = 52). After 12 weeks, subjects were un-blinded but patients were followed for one year. There was a significant difference in the percentage of patients that achieved a 30% reduction in daily visual analog scale scores (p = 0.01). There were significant group differences for all assessments at 12 weeks (p< 0.01).
The most frequent complication of the ONS is lead migration. Various anchoring techniques were described to improve lead stability, however the problem persists.[38] In one review, lead migration was found to be 33% and 60%, 6 months and one year post-implant, respectively. [42] The ONSTIM study reported lead migration in 24% of subjects.[40] The use of self-anchoring leads (e.g. tined leads) looks promising.[37] In a series of 12 patients, only one patient had a few mm lead migration with little change in the stimulation pattern, there was no loss of efficacy.[43] None of 12 patients required a surgical revision for lead migration during a mean follow up period of 13 months (Figure 9). Unlike spinal cord stimulation, wound dehiscence and infection carry a lower morbidity risk although this usually requires total system explant.
C2 Neuralgia
C2 neuralgia is a distinctive type of occipital neuralgia, and it is caused by lesions affecting the C2 nerve root or dorsal ganglion such as neuroma, meningioma, or anomalous vessels.[44-45]
The C2 root lies posterior to the lateral atlanto-axial joint, thus disorders or inflammation of this joint may lead to irritation or entrapment of the nerve root.[46]
C2 neuralgia manifests as intermittent lancinating occipital pain that is associated with lacrimation, ciliary injection, and rhinorrhea. Abolition of pain by selective C2 nerve root block is essential to make the diagnosis. C2 neuralgia that responds poorly to pharmacotherapy and thermocoagulation, decompression, or C2 ganglionectomy may be indicated.[9]
Cervical Muscles
Myofascial Pain
Trigger points in the posterior neck muscles, especially the trapezius, sternocleidomastoid, and the splenius capitis, can generate referred pain in the form of a headache.[47-48]
According to the 3rd edition of the International Classification of Headache Disorders (ICHD), headache causally associated with cervical myofascial tender spots is coded as episodic or chronic tension-type headache associated with pericranial tenderness.[8]
Moreover these tender points usually overlie the zygapophyseal joints, so it is difficult to be distinguished from underlying painful joints.[9]
Needling therapies in the management of myofascial pain showed no efficacy beyond that of placebo.[49] The use of botulinum toxin is controversial. It might be effective in the management of migraine and chronic daily headaches. However, its efficacy in myofascial pain and cervicogenic headaches is debatable.[50-52]
Cervical
Discogenic Pain
C2-C3 provocative discography, but not at the lower levels, can reproduce cervicogenic headache.[53] Radiofrequency lesioning was shown to be effective in obtaining some pain relief for a few months in one study.[54] However, cervical disc interventions are not commonly performed because of the potential for serious complications.
Summary
Cervicogenic headache is one of the most debatable and challenging area in headache medicine. Patients usually benefit the most from a multidisciplinary approach incorporating physical therapy, pharmacotherapy, psychotherapy (biofeedback and relaxation therapy), alternative medicine (acupuncture), and the judicious utilization of interventional pain management modalities.
References
- Sjaastad O, Saunte C, Hovdahl H. et al. Cervicogenic headache. An hypothesis. Cephalalgia 1983; 3: 249-256.
- Fredriksen TA, Hovdal H, Sjaastad O. Cervicogenic headache: clinical manifestation. Cephalalgia 1987; 7: 147-160.
- Sjaastad O. Fredriksen TA, Pfaffenrath V. Cervicogenic headache: diagnostic criteria. Headache 1990; 30: 725-726.
- Leone M, D’Amico D, Moschiano F, et al. Possible identification of cervicogenic headache among patients with migraine: an analysis of 374 headaches. Headache 1995; 35: 461-464.
- Leone M, D’Amico D, Grazzi L, et al. Cervicogenic headache: a critical review of the current diagnostic criteria. Pain 1998; 78: 1-5.
- Suijlekom JA, de Vet HCW, van den Berg SGM, Weber WEJ. Interobserver reliability of diagnostic criteria for cervicogenic headache. Cephalgia 1999; 19: 817-823.
- Sjaastad O. Fredriksen TA, Pfaffenrath V. Cervicogenic headache: diagnostic criteria. Headache 1998; 38: 442-445.
- Headache Classification Committee of the International Headache Society (IHS). The International Classification of Headache Disorders, 3rd edition (beta version). Cephalalgia. 2013;33(9):629-808.
- Bogduk N. The neck and headache. Neurologic clinics 2004;22(1): 151-171.
- Bartsch T, Goadsby PJ. Stimulation of the greater occipital nerve induces increased central excitability of dural afferent input. Brain 2002; 125:1496-1509.
- Bartsch T, Goadsby PJ. Increased responses in trigeminocervical nociceptive neurons to cervical input after stimulation of the dura mater. Brain 2003; 126:1801-1813.
- Narouze S. Algorithms for the diagnosis and management of head and face pain. In: Narouze S, ed. Interventional management of head and face pain: Nerve blocks and beyond. Springer 2014, pp 9-14.
- Aprill C, Axinn MJ, Bogduk N. Occipital headaches stemming from the lateral atlanto-axial (C1-2) joint. Cephalalgia 2002; 22(1):15-22.
- Ehni G, Benner B. occipital neuralgia and the C1-2 arthrosis syndrome. J Neurosurg 1984; 61: 961-965.
- Schonstorm N, Twomey L, taylor J. The lateral atlanto-axial joints and their synovial folds: an in vitro study of soft tissue injury and fractures. J Trauma 1993; 35: 886-892.
- Narouze SN, Casanova J. The efficacy of lateral atlanto-axial intra-articular steroid injection in the management of cervicogenic headache. Anesthesiology 2004; 101: A1005.
- Narouze SN, Casanova J, Mekhail N. The longitudinal effectiveness of lateral atlanto-axial intra-articular steroid injection in the management of cervicogenic headache. Pain Medicine 2007 (in press).
- Joseph B, Kumar B. Gallie’s fusion for atlantoaxial arthrosis with occipital neuralgia. Spine 1994; 19: 454-455.
- Ghanayem AJ, Leventhal M, Bohlman HH. Osteoarthrosis of the atlantoaxial joints- long term follow up after treatment with arthrodesis. J Bone Joint Surg Am 1996; 78: 1300-1307.
- Bogduk N. The clinical anatomy of cervical dorsal rami. Spine 1982; 7: 319-30.
- Lord S, Barnsley L, Wallis B, Bogduk N. Third occipital headache: a prevalence study. J Neurol Neurosurg Psychiatry 1994; 57: 1187-90.
- Lord SM, Barnsley L, Bogduk N. Percutaneous radiofrequency neurotomy in the treatment of cervical zygapophyseal joint pain: a caution. Neurosurgery 1995; 36: 732-39.
- Govind J, King W, Baily B. Bogduk N. Radiofrequency neurotomy for the treatment of third occipital headache. J Neurol Neurosurg Psychiatry 2003; 74: 88-93.
- Slipman CW, Lipetz JS, Plastara CT, et al. Therapeutic zygapophyseal joint injections for headache emanating from the C2-3 joint. Am J Phys Med Rehabil 2001; 80: 182-88.
- Barnsley L, Lord SM, Wallis BJ et al. Lack of effect of intraarticular corticosteroids for chronic pain in the cervical zygapophyseal joints. N Engl J Med. 1994; 330:1047-1050.
- Bovim G, Fredriksen TA, Stolt-Nielsen A, Sjaastad O. Neurolysis of the greater occipital nerve in cervicogenic headache. A follow up study. Headache 1992; 32: 175-9.
- Anthony M. Headache and the greater occipital nerve. Clin Neurol Neurosurg 1992; 94: 297-301.
- van Suijlekom JA, Weber WE, van KM. Cervicogenic headache: techniques of diagnostic nerve blocks. Clin Exp Rheumatol 2000; 18:S39-S44
- Silverman SB. Cervicogenic headache: interventional, anesthetic, and ablative treatment. Curr Pain Headache Rep. 2002; 6: 308-314.
- Sjaastad O, Stolt-Nielsen A, Blume H et al. Cervicogenic headache. Long-term results of radiofrequency treatment of the planum nuchale. Funct Neurol. 1995; 10: 265-271.
- Horowitz MB, Yonas H. Occipital neuralgia treated by intradural dorsal nerve root sectioning. Cephalalgia 1993; 13: 354-60.
- Dubuisson D. Treatment of occipital neuralgia by partial posterior rhizotomy at C1-3. J Neurosurg 1995; 82: 591-6.
- Vanelderen P, Rouwette T, De Vooght P, Puylaert M, Heylen R, Vissers K, Van Zundert J. Pulsed radiofrequency for the treatment of occipital neuralgia: A prospective study with 6 months of followup. Regional Anesthesia and Pain Medicine 2010; 35:148-151.28.
- Choi, H J, Oh IH, Choi SK, Lim YJ. Clinical outcomes of pulsed radiofrequency neuromodulation for the treatment of occipital neuralgia. J Korean Neurosurg Soc 2012; 51:281-285.
- Huang JHY, Galvagno SM Jr, Hameed M, Wilkinson I, Erdek MA, Patel A, Buckenmaier C III, Rosenberg J, Cohen SP. Occipital nerve pulsed radiofrequency treatment: A multi-center study evaluating predictors of outcome. Pain Medicine 2012; 13:489-497.
- Weiner RL, Reed KL. Peripheral neurostimulation for control of intractable occipital neuralgia. Neuromodulation. 1999; 2: 217-221.
- Kapural L, Mekhail N, Hayek SM et al. Occipital nerve electrical stimulation via the midline approach and subcutaneous surgical leads for treatment of severe occipital neuralgia: a pilot study. Anesth Analg. 2005; 101:171-174.
- Weiner RL. Occipital neurostimulation (ONS) for treatment of intractable headache disorders. Pain Med 2006;7:S137-S139.
- Matharu MS, Bartsch T, Ward N, et al. Central neuromodulation in chronic migraine patients with suboccipital stimulation: A PET study. Brain 2004; 127:120-130.
- Saper JR, Dodick DW, Silberstein SD, McCarville S, Sun M, Goadsby PJ. Occipital nerve stimulation for the treatment of intractable chronic migraine headache: ONSTIM feasibility study. Cephalalgia. 2011;31(3):271-285.
- Silberstein SD, Dodick DW, Saper J, et al. Safety and efficacy of peripheral nerve stimulation of the occipital nerves for the management of chronic migraine: results from a randomized, multicenter, double blinded, controlled study. Cephalalgia. 2012;32:1165-1179.
- Schwedt TJ, Dodick DW, Hentz J, Trentman TL, Zimmerman RS. Occipital nerve stimulation for chronic headache- Long term safety and efficacy: Cephalalgia. 2007;27:153-157.
- Narouze SN, Saad R, Nagem H, Kapural L. Occipital Nerve Stimulation with self-anchoring leads for the management of refractory chronic migraine headache (abstract). Pain Med 2009;10:22.
- Kuritzky A. Cluster headache-like pain caused by an upper cervical meningioma. Cephalalgia 1984; 4: 185-6.
- Sharma RR, Parekh HC, Prabhu S, et al. Compression of the C2 root by a rare anomalous ectatic vertebral artery. J Neurosurg 1993; 78: 669-72.
- Poletti CE, Sweet WH. Entrapment of the C2 root and ganglion by the atlantoepistrophic ligament: clinical syndrome and surgical anatomy. Neurosurgery 1990; 27: 288-91.
- Travell J, Rinzler SH. The myofascial genesis of pain. Postgrad Med 1952; 11: 425-34.
- Freund B, Schwartz M. Post-traumatic myofascial pain of the head and neck. Curr Pain Headache Rep. 2002; 6: 361-369.
- Cummings TM, White AR. Needling therapy in the management of myofascial trigger point pain: a systemic review. Arch Phys Med Rehabil 2001; 82: 986-92.
- Cheshire WP, Abashian SW, Mann JD. Botulinum toxin in the treatment of myofascial pain syndrome. Pain 1994; 59: 65-69.
- Freund BJ, Schwartz M. Treatment of whiplash associated neck pain [corrected] with botulinum toxin-A: a pilot study. J Rheumatol. 2000; 27: 481-484.
- Wheeler AH, Goolkasian P, Gretz SS. A randomized, double-blind, prospective pilot study of botulinum toxin injection for refractory, unilateral, cervicothoracic, paraspinal, myofascial pain syndrome. Spine 1998; 23: 1662-1666.
- Grubb SA, Kelly CK. Cervical discography: clinical implications from 12 years of experience. Spine 2000; 25: 1382-1389.
- Blume HG. Cervicogenic headaches: radiofrequency neurotomy and the cervical disc and fusion. Clin Exp Rheumatol. 2000; 18: S53-S58.