Infraclavicular Block
Authors
Jessica J. Buren MD
Instructor
Johnny K. Lee MD
Former Resident Physician
Present Address: Private Practice, Chicago, Illinois
Luminita M. Tureanu MD, FRCPC
Assistant Professor
Department of Anesthesiology
Northwestern University
Chicago, IL
Introduction
Originally, surgical procedures performed on the elbow and distal upper extremity utilizing regional anesthesia were done under axillary block. The axillary block however was not without its limitations. Given the proximal take off of the musculocutaneous nerve, the lateral proximal arm would often be spared. Additionally, the axillary block is difficult to place in patients who are unable to abduct the arm.
The infraclavicular block (ICB) was developed to overcome the limitations of the axillary block. Bazy in 1917, followed by Labat in 1922 are credited with the infraclavicular approach to brachial plexus. In this technique, the needle is directed medially from the midpoint of the clavicle to the anterior tubercle of the transverse process of the sixth cervical vertebra, also known as Chassaignac’s tubercle. This approach however was also not without its own limitations, namely a higher likelihood of pneumothorax as the needle is directed superiorly and medially, near the apex of the lung.
In 1973, Raj reintroduced this approach with some modifications.[1] The aim of his approach was to avoid the positioning limitations and sparing of the musculocutaneous nerve associated with the axillary block, while decreasing the chance of pneumothorax inherent with the Labat approach, by aiming the needle laterally. Since then, several more approaches have been described, each with their own advantages and disadvantages, some of which will be described here.
Advantages
The infraclavicular block has several advantages that make it a preferable approach to brachial plexus blockade: comprehensive upper extremity anesthesia, lower incidence of tourniquet pain and preferable site for catheter insertion.[2,3] The infraclavicular block in general has a higher success rate than the axillary block.[2] ICB has less impact on pulmonary function but is more likely to spare the radial nerve distribution if a single injection is used compared with the supraclavicular approach.[4] The rate of block failure for infraclavicular blocks (11.4%) is similar when compared to other brachial plexus blocks (12.9%). ICB is associated with less tourniquet pain and is less likely to spare the musculocutaneous nerve.[5] There is no difference between the infraclavicular block and all other upper extremity blocks in the observed risk of pneumothorax, vascular puncture, Horner’s syndrome, transient neurological deficit, or systemic local anesthetic toxicity.[2] Block onset time may be slightly longer for infraclavicular block when compared to other single injection brachial plexus blocks.[5] However, when compared to multiple injection axillary block, ICB has a shorter block performance time.[5]
The brachial plexus starts as the union of the ventral primary rami of cervical nerves 5 through 8 (C5-C8) and the thoracic nerve T1 (Figure 1).
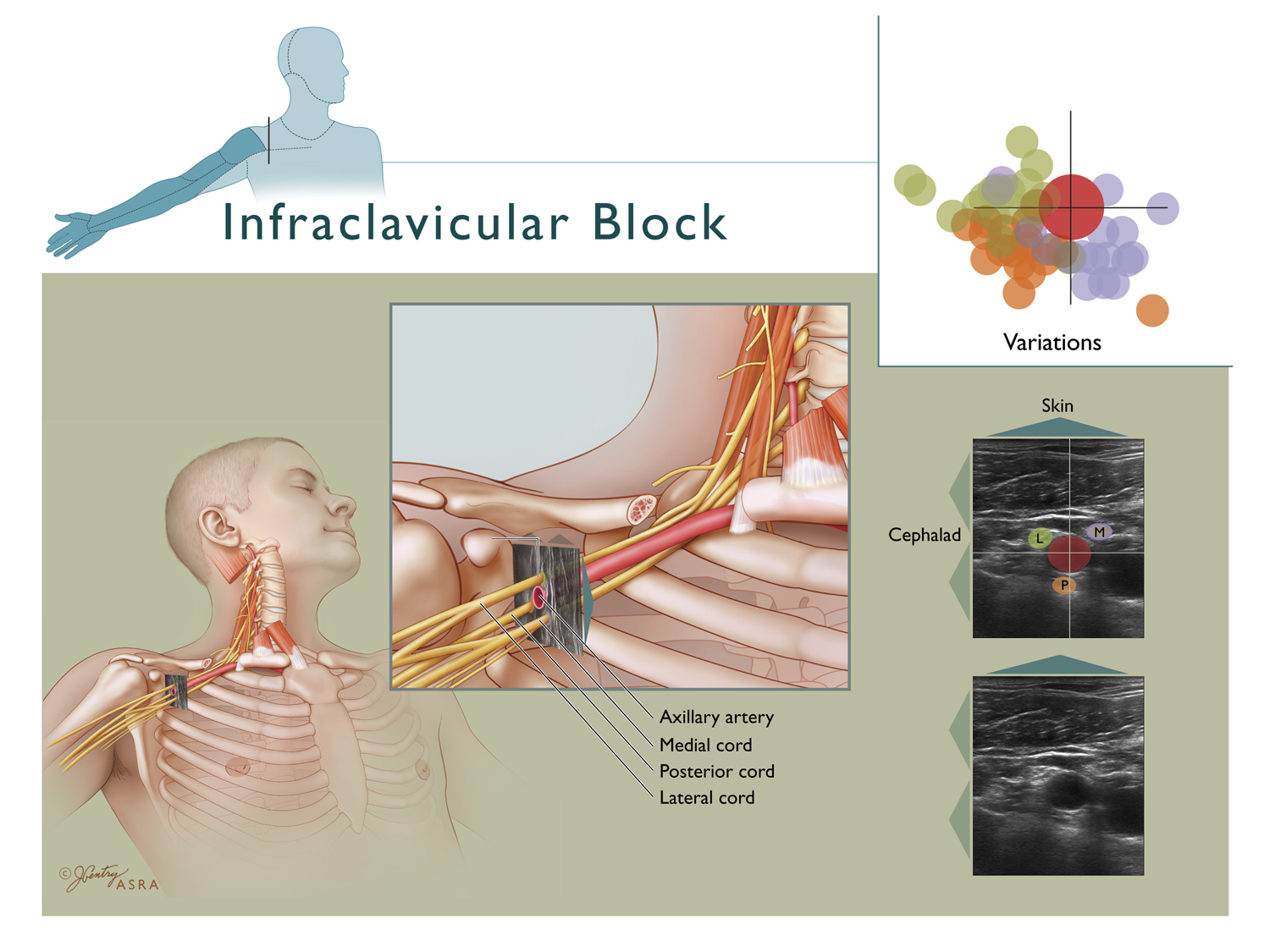
Figure 1. Brachial plexus anatomy.
Copyright 2009 American Society of Regional Anesthesia and Pain Medicine. Used with permission. All rights reserved. Use of this image does not constitute or imply endorsement by ASRA of the website or material on which it appears or the product or service provided.
There are frequent contributions from C4 and T2 as well. The roots of the brachial plexus represent the ventral rami of these spinal nerves. The roots combine above the first rib to form the superior, middle, and inferior trunks of the brachial plexus, between the anterior and middle scalene muscles, in the interscalene groove. As they pass under the clavicle, each trunk divides into an anterior (flexor) and posterior (extensor) division, for a total of six divisions. The divisions undergo yet another level of reorganization into the three cords as the brachial plexus emerges from under the clavicle. These cords are defined by their spatial relationship to the axillary artery, and are designated as the lateral, posterior, and the medial cord. The ICB blocks the brachial plexus at the level of the cords. The branches from the cords go to form the terminal nerves of the upper extremity, namely the musculocutaneous, axillary, median,radial, and ulnar nerves. The point at which the musculocutaneous nerve exits the brachial plexus is important when considering the location at which to block the brachial plexus. A cadaveric study showed that the exit point of the musculocutaneous nerve is typically distal to the coracoid process.[6]
Indications
Indications for the infraclavicular block are the same as those for the axillary and the supraclavicular block, including surgery at the elbow, forearm, wrist, and hand. The infraclavicular block is not suitable for shoulder surgery.
Contraindications
Absolute contraindications for the infraclavicular block include patient refusal or patient’s inability to cooperate, skin infection at the block site, and severe coagulopathy, or allergy to local anesthetic as with any other type of regional anesthetic technique. Severe chronic obstructive pulmonary disease is a relative contraindication for medial approaches given the higher incidence of phrenic nerve paresis. Chest deformity or malunion of previous clavicle fractures that may modify anatomical landmarks may increase the risk of pneumothorax with PNS technique. Other relative contraindications to the infraclavicular block include preexisting active neurologic deficits in the brachial plexus distribution and systemic infection.
Technical Considerations
Surface Anatomy
The infraclavicular block is performed within the infraclavicular fossa which is bordered anteriorly by the pectoralis major and pectoralis minor muscles, superiorly by the distal half of the clavicle, superolaterally by the coracoid process and laterally by the humerus.
Equipment
In general, sterile towels, gauze packs, syringes with local anesthetic, sterile gloves, marking pen, and surface electrode, 8 or 10 cm long, 22 gauge, short bevel, insulated needle, and a peripheral nerve stimulator are used. If an ultrasound guidance technique is used, an ultrasound machine with a high frequency ultrasound probe is needed.
Patient Preparation
In preparation for the procedure, three lead EKG, pulse oximeter, and noninvasive blood pressure monitoring are placed. Sedation and mild analgesia with midazolam 1–5 mg IV and fentanyl 25–100 mcg IV can improve patient comfort and block performance conditions.
The patient is typically positioned with the head of the bed at roughly 30 degrees with the arm slightly abducted. With this position it can be difficult to maneuver and visualize the needle secondary to the close vicinity of the needle insertion point to the clavicle. Auyong et. al. recommended the use of the ‘Houdini clavicle’ position with the shoulder abducted to 90 degrees and the elbow flexed which moves the clavicle cephalad and posterior and allows needle insertion further from the clavicle.[7]
After the skin is aseptically prepared with chlorhexidine solution, sterile towels may be used to drape the area where the block will be performed. The needle insertion site is infiltrated with local anesthetic. The potential discomfort during block needle insertion through the pectoralis muscles can be decreased by infiltrating deeper into the muscle.
Nerve Stimulation Technique
Infraclavicular blocks may be performed using a medial or a lateral approach. The medial approach involves needle insertion on the medial side of the chest wall and vice versa. The medial approaches discussed here are:
- the vertical infraclavicular block;
- the Raj ICB and
- the Borgeat modified ICB.
The lateral approaches discussed are:
- the lateral coracoid approach and
- the lateral sagittal approach.
Medial Approaches
These approaches are performed just caudal to the mid clavicular line. These techniques are technically simpler to perform with peripheral nerve stimulation (PNS) alone. Compared to the lateral approach, needle insertion medially on the chest wall has a higher potential risk of pneumothorax and phrenic nerve paresis.
The vertical infraclavicular block described by Kilka et al in 1995 is a popular block that is simple to perform.[8] With the patient lying supine, the upper extremity to be anesthetized is adducted and flexed at the elbow, while the hand rests on the abdomen. The needle is inserted in an anterior-to-posterior direction, perpendicular to the horizontal plane, just under the clavicle, and at the midpoint between the ventral acromial process of the scapula laterally and the jugular notch of the clavicle.
If the brachial plexus is not identified on the first pass, the needle should be withdrawn and redirected. The needle insertion point is moved 0.5 cm lateral, and if not successful, the needle is moved 0.5 cm medial to the initial insertion point, in the same sagittal plane. It should be kept in mind however that more medial insertion points are associated with a higher incidence of pneumothorax and vascular puncture. Needle advancement greater than 6 cm is not recommended to avoid pneumothorax.[9]
The Raj approach was described in 1973.[1] With this approach, the patient lies supine and faces away from the arm to be blocked. The arm is abducted to 90 degrees and is in neutral position, slightly supinated similar to the position in figure 2.
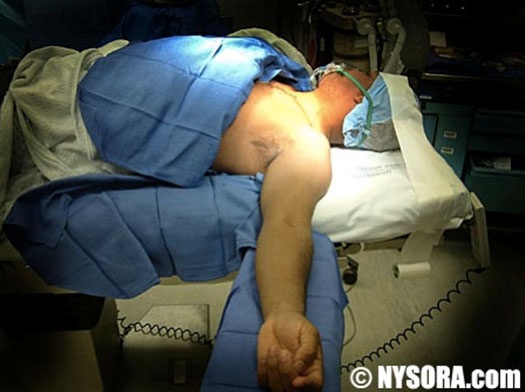
Figure 2. Patient positioning for Raj approach.
From NYSORA.com website, with permission.
Markings are made at the anterior tubercle of the C6 transverse process and the pulsation point of the brachial artery of the arm to be blocked. A line connecting these two points marks the trajectory of the brachial plexus on the skin surface. While standing on the opposite side of the arm to be blocked, the needle is inserted at the midpoint of the inferior border of the clavicle, where the subclavian artery is felt under the clavicle. The needle is inserted through the skin at a 45 degree angle, and directed toward the point where the pulse of the brachial artery is palpated in the arm.
Borgeat Approach (Modified Raj Technique)
Patient positioning is similar to the Raj technique. With the patient lying supine, and facing away from the arm to be blocked, the arm to be anesthetized is abducted to 90 degrees and elevated 30 degrees as in the Raj approach.
A line is drawn connecting the ventral acromial process of the scapula and the jugular notch along the caudal portion of the clavicle (Figure 3). The axillary artery pulsation located in the axillary fossa is also marked.[10]
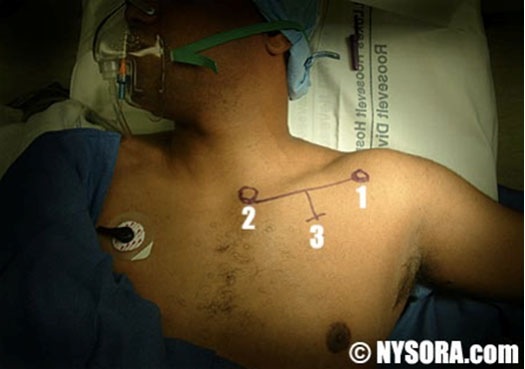
Figure 3. Surface landmarks for the Borgeat approach. 1 = ventral acromial process of the scapula; 2 = the jugular notch of the clavicle and 3 = 1 cm caudal to the clavicle.
Picture taken from NYSORA.com website, with permission.
After local anesthetic skin infiltration, the stimulating needle is inserted 1 cm caudal below the inferior border of the clavicle, in the middle between the acromial process and jugular notch (Figure 4). The needle is directed toward the emergence of the axillary artery in the the axilla, at a 40 to 60 degree angle to the skin. Direct stimulation of the pectoralis muscle indicates that the needle is too shallow and should be advanced. If stimulation of the latissimus dorsi muscle is elicited, the needle is too deep and should be withdrawn and redirected. Elicitation of the deltoid muscle twitch indicates stimulation of the axillary nerve. In this case the needle should be withdrawn and redirected in a more cephalad orientation. Stimulation of the biceps muscle indicates stimulation of the musculocutaneous nerve. In this case, the needle is too cephalad and should be withdrawn and redirected toward a more caudal direction.
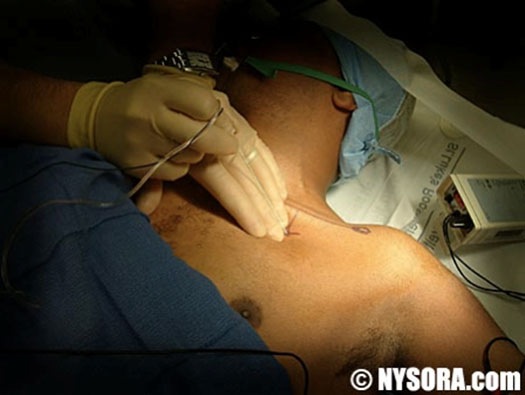
Figure 4. Infraclavicular block using the Borgeat approach
From NYSORA.com website, with permission
Lateral Approaches
Lateral approaches are beneficial as they are less likely to cause hemidiaphragmatic paralysis and pneumothorax since the needle is placed farther away from the pleura. The cords of the brachial plexus, which are named for their position relative to the axillary artery, are however more variable in the lateral location. Additionally, close proximity of the cords to the axillary artery increases the risk of vascular puncture and intravascular injection.
The coracoid approach, described by Whiffler in 1981 underwent several revisions, including one by Wilson in 1998.[11] With the patient lying supine, and the ipsilateral arm abducted or placed next to the trunk, the needle is inserted 2 cm medial and 2 cm caudad to the lateral aspect of the coracoid process. The needle is oriented in the anterior posterior direction, and is usually advanced approximately 5 cm to reach the brachial plexus. In the description of this technique by Wilson et al., the brachial plexus was reached in all 40 patients. Depth needed to reach the plexus was dependent on body habitus, approximately 4.0–4.3 cm.[11]
The advantages cited for the coracoid infraclavicular block are the ability to perform the block in any arm position and the presence of a consistent, palpable bony landmark (coracoid process). In addition, there does not appear to be significant sparing of the musculocutaneous nerve.[12] However, inability to externally compress the site in the case of a hematoma is a disadvantage.
With the patient lying supine, the arm to be blocked is adducted with the hand resting on the abdomen. The patient’s head is slightly elevated and rotated away from the blocked arm.
The operator stands behind the shoulder of the patient and at the head of the bed. The medial point of the coracoid process is palpated. The stimulating needle is inserted at this point, just anterior and caudal to the clavicle at a 0 to 30 degree angle from the skin, aiming caudally. All needle directions should remain in a sagittal plane. In the description of this technique by Klaastad, a 15 degree angle is recommended.[13] A maximum depth of 6.5 cm is recommended to avoid accidental pneumothorax. While inserting the needle, it is recommended to continuously aspirate through the needle to detect puncture of the cephalic vein, axillary artery, or axillary vein.
Nerve Stimulation Endpoints
Several evoked motor responses (EMR) are considered acceptable for the nerve stimulation approach. Flexion of the fingers, extension of hand or fingers, and adduction of the thumb are all acceptable EMR responses at < 0.5 mA, before injection of local anesthetic. Finger flexion implies stimulation of the median nerve component of either the lateral or the medial cord. Extension at the wrist or fingers indicates stimulation of the radial nerve from the posterior cord. Chin et al suggest that posterior cord stimulation is the most reliable indicator of block success.[3] Contraction of the 4th and 5th fingers and thumb adduction indicate stimulation of the ulnar nerve from the medial cord. Although the musculocutaneous nerve is a terminal nerve of the lateral cord, it often exits the lateral cord proximally. Therefore, the stimulation of this nerve is not considered an acceptable EMR, when a single stimulation technique is used.
In single stimulation techniques, data demonstrates improved success rate of the infraclavicular block when local anesthetic is injected after radial nerve stimulation (90%) as compared with the median (74%) or ulnar (68%) evoked motor responses.[14]
A number of studies have demonstrated that injection of more than one cord improves block success, especially when one injection is near the posterior cord. One study demonstrated slightly longer block duration, faster onset time of sensory and motor block, and block success in the multistimulation group of three nerve responses when compared to single stimulation.[15] In a study by Rodriguez et al, similar results were found using the coracoid technique.[16] One study showed that using double stimulation, having initially located and blocked the musculocutaneous nerve, subsequent injection from a radial response resulted in a better success rate with ICB than injection from an ulnar or median response.[17] Rodriguez et al showed that single stimulation of the posterior cord is superior to dual stimulation.[18]
Ultrasound Guided Technique
Ultrasound is an imaging tool for localization of the cords of the brachial plexus in the infraclavicular region. Placement of the ultrasound probe at any position inferior to the clavicle allows visualization of the axillary artery and vein, the surrounding brachial plexus cords, the pleura and pulmonary tissue (Figure 5). A single injection of local anesthetic posterior to the axillary artery was found to have to be superior to a triple injection adjacent to each of the cords.[19] In addition, there is no advantage to double injection over single injection.[20] When local anesthetic is deposited posterior to the axillary artery, one study found a 82.5% ‘anatomical success rate.’ The block failures were because of inadequate spread to all components of the brachial plexus (7.5%) or because most of the local anesthetic spread to the throacoscapular space (10%).[21]
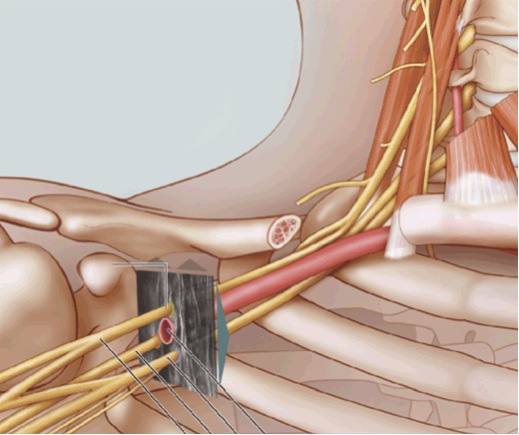
Figure 5. Ultrasound visualization of the brachial plexus cords in the infraclavicular region.
Copyright 2009 American Society of Regional Anesthesia and Pain Medicine. Used with permission. All rights reserved. Use of this image does not constitute or imply endorsement by ASRA of the website or material on which it appears or the product or service provided.
Lateral Sagittal Approach
With the patient lying supine, and the ipsilateral arm in any position, the anesthesiologist performing the procedure stands at the head of the bed, behind the shoulder.
The ultrasound probe is placed in the parasagittal plane medial to the coracoid process and angled to best visualize the axillary artery in cross section, and the three cords (Figure 6).[22] The more medial the placement of the ultrasound probe, the more superficial the pleura will be on the image.
Although the lateral, medial and posterior cords are named for their relative position in relation to the axillary artery, substantial anatomical variation can exist and is best appreciated with the ultrasound. Other vascular structures, such as pectoral vein and arterial branches can be present in between the pectoral muscles, and are easily identified with color Doppler.
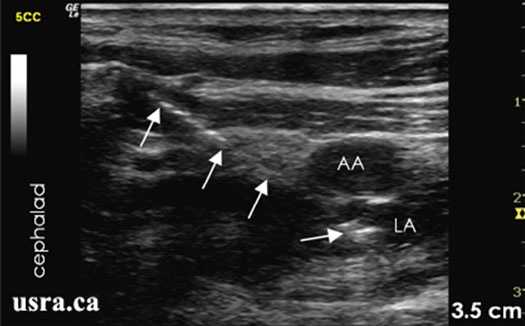
Figure 6. Sonoanatomy of the infraclavicular region showing needle advancement using an in plane lateral approach. Arrows = block needle; AA = axillary artery; LA = local anesthetic; note needle and local anesthetic posterior to artery.
From usra.ca website, with permission.
Vertical Infraclavicular Approach
A vertical infraclavicular in-plane or out-of-plane approach has also been described under US guidance. For this approach the ultrasound probe is positioned under and parallel to the clavicle and the needle insertion point is the middle of clavicle. The brachial plexus is more superficial in this location and is visualized lateral to the subclavian artery.[23]
The stimulating needle is inserted immediately next to the ultrasound probe at approximately 60–70° angle. An in-plane approach is recommended for continuous direct visualization of the needle under ultrasound (Figure 7). Injection of local anesthetic lateral to the subclavian artery and especially close to the posterior cord, with a U shape spread of the injectate is most often associated with a reliable block (Figure 8). A dual technique, using PNS in conjunction with US may be used if needed.
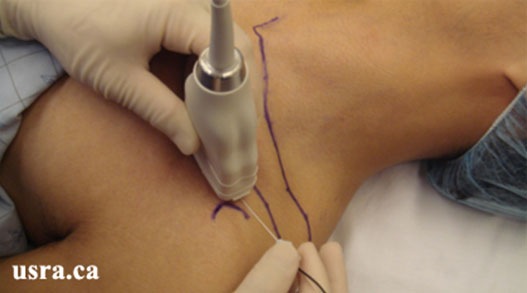
Figure 7. In-plane needle technique for the vertical infraclavicular approach.
From usra.ca website, with permission.
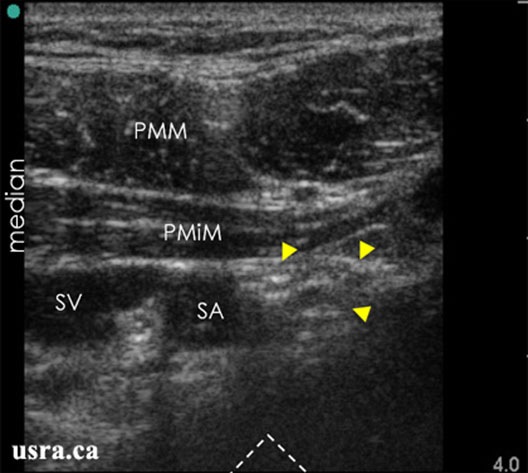
Figure 8. Sonoanatomy of the infraclavicular region with the transducer placed transverse below the clavicle; arrowheads = nerves; PMM & PMiM = pectoralis major & minor muscles; SA & SV = subclavian artery & vein.
From usra.ca website, with permission.
Nerve Stimulation Versus Ultrasound Guidance
Nerve stimulation techniques have been well described for many years. However ultrasound (US) is an emerging technology and is quickly gaining acceptance. There are several recognized advantages of ultrasound guidance as a nerve localization technique: direct visualization of the nerve, surrounding anatomy, including vascular structures, and spread of local anesthetic injectate. In general, studies have demonstrated quicker onset of sensory and motor blockade and longer duration with US guidance compared to peripheral nerve stimulation (PNS) alone.[24-26] US guidance is also associated with fewer needle passes and less patient discomfort during block performance.[24] Eren et al found that they could also reduce the doses of local anesthetic to 70% of those required by nerve stimulator techniques.[27]
There is current level Ib, grade A evidence that US guidance of upper extremity blocks results in faster onset of sensory block and higher success rates.[28] Sauter et al in a study comparing electrical nerve stimulation and ultrasound guided lateral sagittal infraclavicular blocks, did not demonstrate differences in the time until readiness for surgery, discomfort associated with the block procedure, nor pain related to tourniquet ischemia.[29] The success rate for blocks aided by ultrasound was 95% versus 85% for blocks with PNS, not statistically significant. However, the sample size of the study was not powered to demonstrate this difference.[29] More recent studies showed that US guided ICB reduces block performance time and improves readiness for surgery.[30-31]
Currently, there is no RCT data regarding improved safety outcomes with US guidance for peripheral nerve blocks, as intravascular injection and nerve injury can still occur with PNS and US guidance as well.[32-33] US is nevertheless a sensitive indicator of needle to nerve contact, more sensitive than paresthesia or PNS. US guidance is also recognized for its ability to reduce the likelihood of unintended vascular puncture, and to facilitate a reduction of the volume of injected local anesthetic.[34]
Dual Ultrasound and Peripheral Nerve Stimulation Technique
To date, the combination of nerve stimulation and US guidance does not seem to add value to ultrasound guidance alone. In a study of US guided interscalene blockade, the addition of nerve stimulation had no impact on success or duration of blockade.[35]
Specifically to ICB, one study demonstrated shorter performance time for the ICB with ultrasound guidance alone when compared with ultrasound guidance plus peripheral nerve stimulation. Visualization of local anesthetic spread posterior-medio-lateral to the axillary artery reliably produced complete brachial plexus block.[36]
Continuous Infraclavicular Block
When prolonged postoperative analgesia is desired, placement of a perineural infraclavicular catheter is appropriate. The infraclavicular location offers several advantages, such as a lower risk of dislodgement, easy to inspect and maintain the catheter site and easy to secure the catheter to the anterior chest. In addition, Mariano et al found that infraclavicular continuous catheters provided superior analgesia when compared to supraclavicular continuous catheters.[37]
Continuous perineural catheters for orthopedic surgeries have emerged as a useful technique for post operative pain control and decreased time to discharge readiness.[38-39] Compared to single shot technique, continuous infraclavicular block has the benefits of decreased sleep disturbances, decreased opioid use and associated side effects, with overall improvement of pain management satisfaction.[40] Continuous infraclavicular catheter techniques have been described utilizing peripheral nerve stimulation and ultrasound guidance. In a recent study, Mariano et al showed that placement of perineural infraclavicular catheters under ultrasound guidance has several advantages over PNS: higher success rate, shorter procedure time and fewer inadvertent vascular punctures.[41]
Until recently, there seemed to be no conclusive evidence of benefits of stimulating catheters over non stimulating catheters.[42-43] However, a recent systematic review of 12 RCTs showed that usage of stimulating catheters may lead to better analgesic quality.[44] One study specifically comparing ICB catheters showed a higher block success using stimulating catheters and ultrasound guidance compared to placement of either stimulating or non-stimulating catheter without ultrasound.[45]
Regarding pump infusion strategies, a continuous infusion rate in combination with patient-controlled bolus doses via an infraclavicular perineural catheter improved analgesia and decreased opioid use when compared with continuous only or bolus only strategies alone.[46]
Technical Considerations
After standard monitors are applied, the patient is sedated as needed and positioned, the skin is prepared as usual with 2% chlorhexidine in 70% alcohol or betadine solution. A sterile drape is placed around the location where the catheter will be placed and anchored. An ultrasound transducer is placed into a sterile sheath with acoustic gel and secured with rubber band around the transducer head. A 17G or 18 G insulated Tuohy needle is used to contact the neural structures.
Procedure
The coracoid approach has been described for placement of continuous infraclavicular block. However difficulty may arise as the needle and the brachial plexus intercept one another at a 90 degree angle, and thus advancing the catheter may be problematic.[40]
If an US-guided technique is used, with an in an in plane approach, the needle is inserted and advanced to reach the posterior aspect of the axillary artery (indicated by the arrow in Figures 9 and 10). Injection of a small amount of local anesthetic or D5W may be used to distend the perineural space and facilitate threading of the catheter. The catheter, simulating or non stimulating, would be advanced 3-5 cm perineural Afterwards, local anesthetic is injected through the catheter and spread of injectate observed under real time US guidance. The catheter may be tunneled if needed, and secured at the skin using steri strips and Tegaderm.
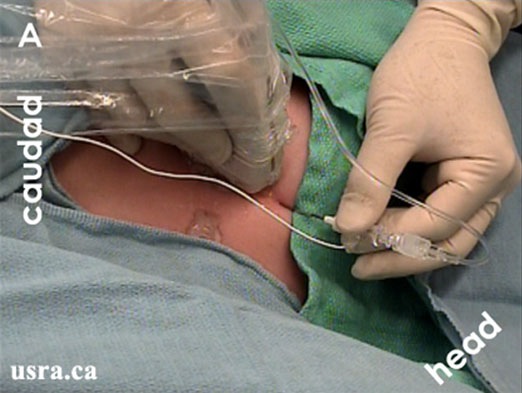
Figure 9. In plane approach with stimulating Tuohy needle for continuous perineural catheter placement.
From usra.ca website, with permission
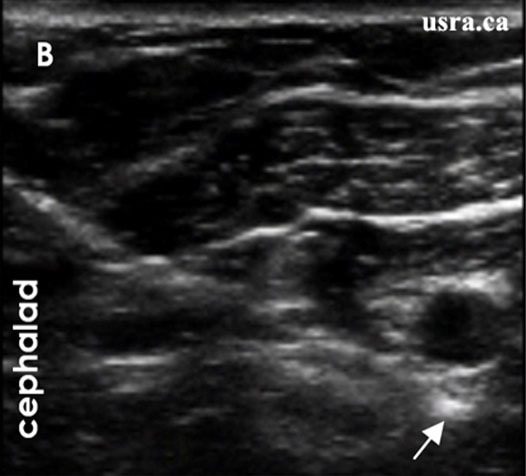
Figure 10. Sonogram showing needle tip posterior to the axillary artery.
From usra.ca website, with permission.
An out of plane technique can also be used for placement of the catheter.
Among the different approaches of infraclavicular block, the vertical infraclavicular approach and its modifications has the highest success rate with PNS alone. Use of ultrasound improves the success of the lateral sagittal approach to that similar to the vertical approach.[47]
Choice of Local Anesthetic
Choice of local anesthetic dose, concentration and volume depends on the indication of the infraclavicular block and the need for surgical anesthesia and postoperative analgesia. Factors to be considered are: type and duration of surgery, speed of onset, single shot versus continuous catheter, inpatient versus outpatient, protection of the insensate limb, and postoperative neurological examination.
Both mepivacaine 1.5% and lidocaine 2% have quick onset and intermediate duration of action (3-6 hours of anesthesia and up to 8 hours of analgesia). Generally 0.75% ropivacaine and 0.5% bupivacaine are considered equipotent and provide excellent surgical anesthesia, with an onset time of 15-20 min and analgesia lasting 8-12 hours. Adding mepivicaine to bupivacaine has been found to reduce the block onset time when compared to bupivicaine alone.[48] Ropivacaine 0.5% is also suitable for surgical anesthesia, while lower concentrations (e.g., 0.2%) may be used for analgesia. The infraclavicular block generally is considered a high volume block and requires 30-40 mL of local anesthetic for complete anesthesia during single shot injection. For ropivicaine 0.75% in particular, one study found that 31 mL was the minimum effective volume for 95% of patients.[49]
For continuous ICB, 0.2% ropivacaine is indicated. After a 10–15 mL bolus injection through the catheter, an infusion may be started at 8 mL/h. A lower rate (e.g., 5mL/h) may be indicated if a patient controlled bolus option is added. An on demand 3-5 mL bolus every 30 minutes is appropriate for breakthrough pain. One study showed that lower infusion rate with higher concentration of local anesthetic increased the chance of insensate extremities, and thus the possibility of accidental injury. As such, a higher basal infusion rate with a lower concentration of local anesthetic is recommended.[50]
Complications
The infraclavicular block is subject to all general complications associated with any brachial plexus block. These complications include, but are not limited to, vascular injury and hematoma, infection, local anesthetic toxicity from either direct intravascular injection or systemic absorption resulting in seizures or cardiac arrest, primary block failure or secondary block failure (with perineural catheters), nerve injury, and even death.
Complications more specific to the infraclavicular block include pneumothorax and hemi-diaphragmatic paresis. As noted before, lateral sagittal and coracoid approaches seem to carry less risk than the medial approaches (e.g., vertical infraclavicular block). However, pneumothorax is rare even with the vertical infraclavicular block, which reportedly rate of about 0.2 to 0.7%.[51] There have been case reports of pneumothorax even with lateral approaches however.[52] It is possible that the use of ultrasound may further decrease this risk.[53] There have been reports of hemidiaphragmatic paresis with ultrasound guided lateral sagittal ICB and and pericoracoid approaches.[54-55] In a large retrospective study on 1146 patients who received an US guided ICB, the success rate was 99.3%, with no reported cases of nerve injury, pneumothorax, or local anesthetic toxicity. The incidence of asymptomatic arterial puncture was 0.7%.[56] However, there has been a case of Horner’s syndrome reported after an ultrasound guided infraclavicular block.[57]
Summary
The infraclavicular block is a simple and easy block to perform, and an effective alternative to supraclavicular or axillary block for upper extremity surgery, except for shoulder surgery. One major advantage is that there is no need for arm abduction while positioning the patient. The incidence of pneumothorax and hemi-diaphragmatic paralysis is less that with other brachial plexus approaches. Continuous perineural catheters can be safely secured to the upper chest wall. Ultrasound guidance has been shown to improve performance time, success rate and a decrease risk of associated complications.
References
- Raj PP, Montgomery SJ, Nettles D, et al. Infraclavicular brachial plexus block—a new approach. Anesth Analg 1973;52:897–904.
- Chin KJ, Singh M, Velayutham V, et al. Infraclavicular brachial plexus block for regional anaesthesia of the lower arm. Cochrane Database Syst Rev. 2010;2:CD005487
- Chin KJ, Singh M, Velayutham V, Chee V. Infraclavicular brachial plexus block for regional anaesthesia of the lower arm. Anesth Analg. 2010;111:1072.
- Arcand G, Williams SR, Chouinard P, et al. Ultrasound-guided infraclavicular block versus supraclavicular block. Anesth Analg 2005;101:886-90.
- Chin KJ, Alakkad H, Adhikary SD, Singh M. Infraclavicular brachial plexus block for regional anaesthesia of the lower arm. Cochrane Database Syst Rev. 2013 Aug 28;8:CD005487.
- Pianezza A, Salces y Nedeo A, Chaynes P, Bickler PE, Minville V. The emergence level of the musculocutaneous nerve from the brachial plexus: implications for infraclavicular nerve blocks. Anesth Analg. 2012;114:1131-3.
- Auyong DB, Gonzales J, Benonis JG. The Houdini clavicle: arm abduction and needle insertion site adjustment improves needle visibility for the infraclavicular nerve block. Reg Anesth Pain Med. 2010;35:403-4.
- Kilka HG, Geiger P, Mehrkens HH. Infraclavicular vertical brachial plexus blockade. A new method for anesthesia of the upper extremity. An anatomical and clinical study. Anaesthesist 1995;44:339–44
- Klaastad O, Smedby O, Kjelstrup T, et al. The vertical infraclavicular brachial plexus block: a simulation study using magnetic resonance imaging. Anesth Analg 2005;101:273–8
- Borgeat A, Ekatodramis G, Dumont C. An evaluation of the infraclavicular block via a modified approach of the Raj technique. Anesth Analg. 2001;93:436-44
- Wilson JL, Brown DL, Wong GY. Infraclavicular brachial plexus block: parasagittal anatomy important to the coracoid technique. Anesth Analg. 1998;87:870-3
- Mosaffa F, Gharaei B, Rafeeyan M, Gachkar L. Comparing vertical and coracoid approaches for infraclavicular block in orthopedic surgery of the forearm and hand. J Clin Anesth. 2012;24:196-200.
- Klaastad Ø, Smith HJ, Smedby O, et al. A novel infraclavicular brachial plexus block: the lateral and sagittal technique, developed by magnetic resonance imaging studies. Anesth Analg. 2004;98:252-6
- Bloc S, Garnier T, Komly B, et al. Single-stimulation, low-volume infraclavicular plexus block: influence of the evoked distal motor response on success rate. Reg Anesth Pain Med 2006;31:433–7
- Gaertner E, Estebe JP, Zamfir A, et al. Infraclavicular plexus block: Multiple injections versus single injection. Reg Anesth Pain Med 2002;27:590-4
- Rodriguez J, Barcena M, Taboada-Muniz M, et al. A comparison of single versus multiple injections on the extent of anesthesia with coracoid infraclavicular brachial plexus block. Anesth Analg 2004;99:1225-30.
- Minville V, Fourcade O, Bourdet B, et al. The optimal motor response for infraclavicular brachial plexus block. Anesth Analg 2007;104:448-51
- Rodríguez J, Taboada M, Oliveira J, et al.Single stimulation of the posterior cord is superior to dual nerve stimulation in a coracoid block.Acta Anaesthesiol Scand 2010;54:241-5.
- Fredrickson MJ, Wolstencroft P, Kejriwal R, Yoon A, Boland MR, Chinchanwala S. Single versus triple injection ultrasound-guided infraclavicular block: confirmation of the effectiveness of the single injection technique. Anesth Analg. 2010;111:1325-7.
- De Tran QH, Bertini P, Zaouter C, Muñoz L, Finlayson RJ. A prospective, randomized comparison between single- and double-injection ultrasound-guided infraclavicular brachial plexus block. Reg Anesth Pain Med. 2010;35:16-21.
- Benkhadra M, Faust A, Fournier R, Aho LS, Girard C, Feigl G. Possible explanation for failures during infraclavicular block: an anatomical observation on Thiel's embalmed cadavers. Br J Anaesth. 2012;109:128-9.
- Sandhu NS,Capan LM. Ultrasound-guided infraclavicular brachial plexus block. Br J Anaesth. 2002;89:254-9.
- Ootaki C,Hayashi H,Amano M. Ultrasound-guided infraclavicular brachial plexus block: an alternative technique to anatomical landmark-guided approaches. Reg Anesth Pain Med 2000;25:600-4.
- Casati A, Danelli G, Baciarello M, et al. A prospective, randomized comparison between ultrasound and nerve stimulation guidance for multiple injection axillary brachial plexus block. Anesthesiology 2007;106:992-6.
- Marhofer P, Sitzwohl C, Greher M, et al. Ultrasound guidance for infraclavicular brachial plexus anaesthesia in children. Anaesthesia 2004;59:642-64
- Trabelsi W, Amor MB, Lebbi MA, Romdhani C, Dhahri S, Ferjani M. Ultrasound does not shorten the duration of procedure but provides a faster sensory and motor block onset in comparison to nerve stimulator in infraclavicular brachial plexus block. Korean J Anesthesiol. 2013;64:327-33.
- Eren G, Altun E, Pektas Y, Polat Y, Cetingok H, Demir G, Bilgi D, Tekdos Y, Dogan M. To what extent can local anesthetics be reduced for infraclavicular block with ultrasound guidance? Anaesthesist. 2014 Aug 8. [Epub ahead of print]
- Neal JM, Brull R, Chan VW, et al.The ASRA evidence-based medicine assessment of ultrasound-guided regional anesthesia and pain medicine: Executive summary.Reg Anesth Pain Med 2010;35:S1-S9.
- Sauter AR, Dodgson MS, Stubhaug A, et al. Electrical nerve stimulation or ultrasound guidance for lateral sagittal infraclavicular blocks: a randomized, controlled, observer-blinded, comparative study. Anesth Analg 2008;106:1910-15.
- Brull R, Lupu M, Perlas A, et al. Compared with dual nerve stimulation, ultrasound guidance shortens the time for infraclavicular block performance.Can J Anaesth. 2009;56:812-8.
- Taboada M, Rodríguez J, Amor M, et al.Is ultrasound guidance superior to conventional nerve stimulation for coracoid infraclavicular brachial plexus block?Reg Anesth Pain Med 2009;34:357-60.
- Hebl JR. Ultrasound-guided regional anesthesia and the prevention of neurologic injury: fact or fiction? Anesthesiology 2008; 108:186-8.
- Zetlaoui RJ, Labbe J-P, Benhamou D. Ultrasound guidance for axillary plexus block does not prevent intravascular injection. Anesthesiology 2008;108:761.
- Neal JM. Ultrasound guided regional anesthesia and patient safety. Reg Anesth Pain Med 2010;35:S57-67.
- Sinha SK, Abrams JH, Weller RS. Ultrasound-guided interscalene needle placement produces successful anesthesia regardless of motor stimulation above or below 0.5 mA. Anesth Analg. 2007;105:848-52.
- Dingemans E, Williams SR, Arcand G, et al. Neurostimulation in ultrasound-guided infraclavicular block: a prospective randomized trial. Anesth Analg. 2007;104:1275-80.
- Mariano ER, Sandhu NS, Loland VJ, Bishop ML, Madison SJ, Abrams RA, Meunier MJ, Ferguson EJ, Ilfeld BM. A randomized comparison of infraclavicular and supraclavicular continuous peripheral nerve blocks for postoperative analgesia. Reg Anesth Pain Med. 2011;36:26-31.
- Ilfeld BM, Vandenborne K, Duncan PW, et al. Ambulatory continuous interscalene nerve blocks decrease the time to discharge readiness after total shoulder arthroplasty. A randomized, triple-masked, placebo-controlled study. Anesthesiology. 2006;105:999-1007.
- Hebl JR, Dilger JA, Byer DE, et al. A pre-emptive multimodal pathway featuring peripheral nerve block improves perioperative outcomes after major orthopedic surgery. Reg Anesth Pain Med. 2008;33:510-17.
- Ilfeld BM, Morey TE, Enneking FK. Continuous infraclavicular brachial plexus block for postoperative pain control at home: a randomized, double-blinded, placebo-controlled study. Anesthesiology. 2002;96:1297-1304.
- Mariano ER, Loland VJ, Bellars RH, et al. Ultrasound guidance versus electrical stimulation for infraclavicular brachial plexus perineural catheter insertion. J Ultrasound Med 2009;28:1211-8.
- Birnbaum J, Kip M, Spies CD, et al. The effect of stimulating versus non-stimulating catheters for continuous interscalene plexus blocks in short-term pain management. J Clin Anesth. 2007;19:434-39.
- Tran DQH, Munoz L, Russo G, et al. Ultrasonography and stimulating perineural catheters for nerve blocks: a review of the evidence. Can J Anaesth 2008;55:447-57.
- Morin AM, Kranke P, Wulf H, et al. The effect of stimulating versus nonstimulating catheter techniques for continuous regional anesthesia: a semiquantitative systematic review. Reg Anesth Pain Med 2010;35:194-9.
- Dhir S, Ganapathy S. Comparative evaluation of ultrasound-guided continuous infraclavicular brachial plexus block with stimulating catheter and traditional technique: a prospective- randomized trial. Acta Anaesthesiol Scand 2008;52:1158-66.
- Ilfeld BM, Morey TE, Enneking FK. Infraclavicular perineural local anesthetic infusion: a comparison of three dosing regimens for postoperative analgesia. Anesthesiology 2004;100:395-402
- Macfarlane A., Anderson, K. Infraclavicular brachial plexus blocks. Continuing Education in Anaesthesia, Critical Care & Pain 2009;9:139-43.
- Laur JJ, Bayman EO, Foldes PJ, Rosenquist RW. Triple-blind randomized clinical trial of time until sensory change using 1.5% mepivacaine with epinephrine, 0.5% bupivacaine, or an equal mixture of both for infraclavicular block. Reg Anesth Pain Med. 2012;37:28-33.
- Flohr-Madsen S, Ytrebø LM, Kregnes S, Wilsgaard T, Klaastad Ø. Minimum effective volume of ropivacaine 7.5 mg/ml for an ultrasound-guided infraclavicular brachial plexus block. Acta Anaesthesiol Scand. 2013;57:495-501.
- Ilfeld BM, Le LT, Ramjohn J, et al. The effects of local anesthetic concentration and dose on continuous infraclavicular nerve blocks: a multicenter, randomized, observer masked, controlled study. Anesth Analg 2009;108:345–50.
- Neuburger M, Landes H, Kaiser H. Pneumothorax in vertical infraclavicular block of the brachial plexus. Review of a rare complication. Anaesthetist 2000;49:901–4.
- Crews JC, Gerancher JC, Weller RS. Pneumothorax after coracoid infraclavicular brachial plexus block. Anesth Analg 2007;105:275-7.
- Gauss A, Tugtekin I, Georgieff M, Dinse-Lambracht A, Keipke D, Gorsewski G. Incidence of clinically symptomatic pneumothorax in ultrasound-guided infraclavicular and supraclavicular brachial plexus block. Anaesthesia. 2014;69:327-36.
- Yang CW, Jung SM, Kwon HU, Kang PS, Cho CK, Choi HJ. Transient hemidiaphragmatic paresis after ultrasound-guided lateral sagittal infraclavicular block. J Clin Anesth. 2013;25:496-8.
- Lee HS, Lee JH, Cha YD, Park SK, Seo CH, Han JU Phrenic nerve palsy following infraclavicular brachial plexus block. Korean J Anesthesiol 2009;57:515-7.
- Sandhu NS, Manne JS, Medabalmi PK, et al. Sonographically guided infraclavicular brachial plexus block in adults: a retrospective analysis of 1146 cases. J Ultrasound Med 2006;25:1555-61
- Walid T, Mondher BA, Mohamed Anis L, Mustapha F. A Case of Horner's Syndrome following Ultrasound-Guided Infraclavicular Brachial Plexus Block. Case Rep Anesthesiol. 2012;2012:125346