How I Do It: Erector Spinae Plane Block and Spinal Anesthesia for Same Day Lumbar Spine Surgery
Cite as: Deloso E, Bojaxhi E. How I do it: erector spinae plane block and spinal anesthesia for same day lumbar spine surgery. ASRA Pain Medicine News 2024;49. https://doi.org/10.52211/asra020124.011.
Several studies have supported the benefits of using spinal anesthesia (SA) over general anesthesia (GA) in some lumbar spine surgeries. The use of SA rather than GA for lumbar spine surgery is associated with similar hypotension,1,2 less postoperative nausea and vomiting (PONV),1,3,4 decreased blood loss,1,3,5,6 shorter total operative time,1,5,6 and a shorter hospital length of stay.1,6 To date, there have been no reports of increased risk of neurologic complications associated with SA versus GA in lumbar spine surgery.1,3 The erector spinae plane block for lumbar spine surgery under GA has demonstrated improved postoperative pain control, decreased opioid consumption, and reduced PONV.7–9
This article describes how we employ SA for surgical anesthesia combined with a preoperative erector spinae plane (ESP) block for postoperative pain control in non-complex lumbar spine surgeries. Our technique has significantly reduced our hospital length of stay and reliably allows for same-day discharge for elective lumbar spine surgeries.
Patient Selection
The option of awake spine surgery is presented to the patient by the neurosurgeon during the presurgical consultation. Selection criteria include:
- The patient elects to undergo the surgery under a spinal anesthetic.
- No history or anticipation of a difficult airway.
- The patient is comfortable in the prone position.
- Simple 1-2 level lumbar surgery.
- Procedure duration less than 150 minutes.
- Body Mass Index less than 35.
- Full neck/shoulder mobility.
Perioperative Course
Multimodal analgesia and antiemetics are employed, including preoperative administration of 1,000 mg PO acetaminophen, 400 mg PO celecoxib, 40 mg PO aprepitant, and 20 mg PO famotidine, followed by intraoperative administration of 8 mg IV dexamethasone and 4 mg IV ondansetron.
In the preoperative area, a bilateral erector spinae block is performed with 10 ml of 0.5% bupivacaine on each side, one vertebral level above the surgical intervention (Figure 1). A sensory examination is then conducted to confirm the success of the block.
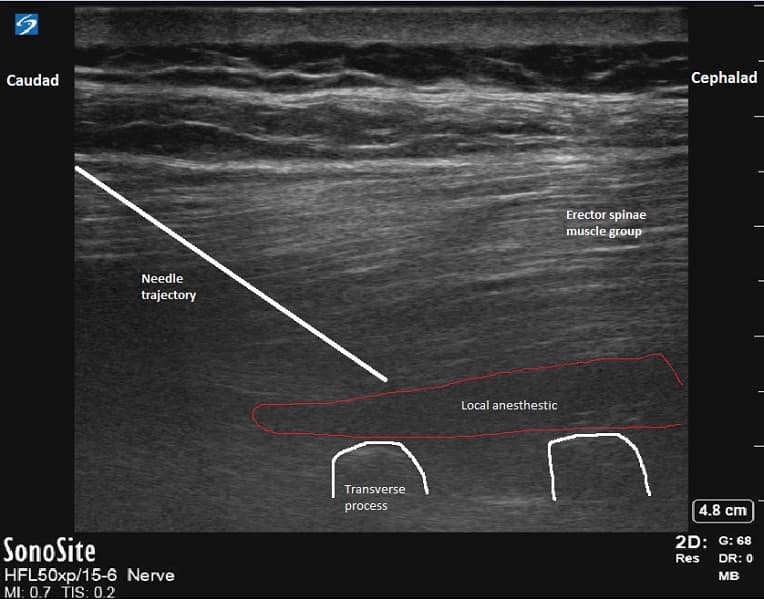
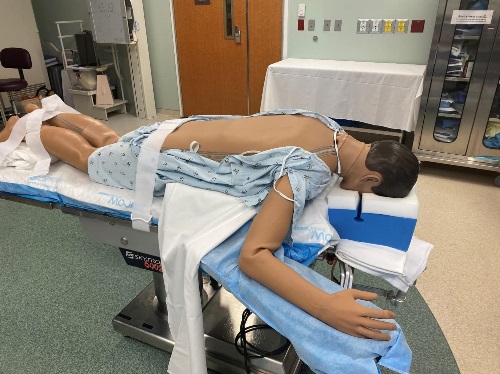
Upon arrival at the operating room, sedation is initiated, aiming to maintain the patient's ability to comprehend and follow commands while ensuring comfort during the prone positioning after the spinal injection. We use dexmedetomidine at a dosage of 0.1 – 0.5 mcg/kg/hr, and if needed, an anxiolysis bolus of 20-30 mg of propofol. The spinal injection is performed as a single shot on the transport stretcher using 12.5 mg of isobaric, preservative-free 0.5% bupivacaine. Once the spinal injection is successfully administered, the patient is positioned comfortably prone on a Wilson frame with a foam headrest (Figure 2). To enhance patient comfort, noise-canceling headphones playing music of the patient's choice are offered
Intraoperative Troubleshooting
If the patient requires supplemental anxiolysis, a low-dose propofol infusion (20-30 mcg/kg/min) can be started. It is essential to exercise caution when titrating additional sedatives to prevent the patient from becoming unresponsive to verbal stimulation. Oversedation may lead to a disinhibited, disoriented, and uncooperative patient, which can impede surgical progression.
Occasionally, the patient may experience discomfort for which intraoperative local anesthetic infiltration may be needed. This discomfort may be described as pressure due to retracting or pulling of the soft tissue during surgical exposure. It can occur despite a successful spinal anesthetic, and in such cases, the surgeon may inject 5-10 cc of 2% lidocaine into the soft tissue. However, our practice has shown that the addition of a pre-surgical ESP block has made the need for the surgeon to inject additional local anesthetic extremely rare.
While scheduling a long procedure under spinal anesthesia should be avoided, it is possible for an elective surgery to continue longer than expected. If the patient starts reporting discomfort due to the regression of the spinal anesthetic's effect, the neurosurgical and anesthesia teams need to have a comprehensive plan. If the spinal anesthesia is starting to wear off during closure, local anesthetic injection into the skin incision may be helpful. However, if osseous manipulation remains a part of the procedure, the spinal anesthetic needs to be supplemented, or the case should be converted to general anesthesia. Spinal anesthetic supplementation can be performed by instructing the neurosurgical team to inject 2.5 mg (0.5 mL) of 0.5% bupivacaine intrathecally with a tuberculin syringe.
Our technique has significantly reduced our hospital length of stay and reliably allows for same-day discharge for elective lumbar spine surgeries.
In extreme discomfort or emergency situations, patients may need to be converted to general anesthesia. Emergent conversions would require a timely and coordinated effort, best rehearsed in multidisciplinary simulations. Our process of emergently converting to general anesthesia due to airway compromise includes calling for additional anesthesia help, preparing to flip the patient from prone to supine, and attempting to place a laryngeal mask airway (LMA) while prone. If the first attempt at a prone LMA is unsuccessful, the patient should be repositioned supine. The repositioning process should have already begun with the OR nurse bringing the stretcher into the OR and the surgical team removing protruding instruments and applying sterile dressing to the surgical site. After flipping and securing the airway, there should be an interdisciplinary discussion on whether to proceed with the surgery. Fortunately, with careful patient selection and multidisciplinary coordinated care, we have yet to convert a case to general anesthesia during the surgery.
Post-Op
Sedation is discontinued at the time of closing. Once in the post-anesthesia care unit (PACU), patients promptly receive the multimodal oral pain and antinausea regimen they will be prescribed upon discharge: acetaminophen 1000 mg PO every 6 hours, methocarbamol 1000-1500 mg PO every 8 hours, oxycodone 5-10 mg PO every 4 hours, and sublingual ondansetron. Additionally, the diet is advanced as quickly as tolerated. Early ambulation with assistance is facilitated, and patients are assisted to a chair once they regain lower extremity strength.
Patients are considered ready to be discharged home from the PACU if they can walk and if their pain and nausea are under control with the prescribed home regimen. If patients are unable to meet these criteria, they remain as inpatients for a 23-hour observation period.
Conclusion
In conclusion, the protocol incorporating spinal anesthesia and an erector spinae plane block for spine surgery demonstrates numerous advantages. It improves patient comfort,7–9 reduces post-operative fatigue,11 and increases overall patient satisfaction.4,7,8 At the same time, it optimizes healthcare utilization,5,6 reduces hospital stays,1,5–9 shortens operative times,2,5,6 and enhances cost-effectiveness for healthcare facilities. 5,6 This comprehensive approach to spine surgery reflects a patient-centric focus while also contributing to more efficient and resource-conscious healthcare practices.


References
- Urick D, Sciavolino B, Wang TY, et al. Perioperative outcomes of general versus spinal anesthesia in the lumbar spine surgery population: a systematic review and metaanalysis of data from 2005 through 2021. J Clin Orthop Trauma 2022;16:30:101923. https://doi.org/10.1016/j.jcot.2022.101923
- De Biase G, Gruenbaum SE, West JL, et al. Spinal versus general anesthesia for minimally invasive transforaminal lumbar interbody fusion: implications on operating room time, pain, and ambulation. Neurosurg Focus 2021;51(6):E3. https://doi.org/10.3171/2021.9.FOCUS21265
- Borden S, Flores RA. Neuraxial anesthesia versus general anesthesia in spine surgery patients: benefits, risks, and why it should be considered. ASRA Pain Medicine News 2018;43. https://www.asra.com/news-publications/asra-newsletter/newsletter-item/asra-news/2018/07/20/neuraxial-anesthesia-versus-general-anesthesia-in-spine-surgery-patients-benefits-risks-and-why-it-should-be-considered
- Oh SK, Lim BG, Won YJ, et al. Analgesic efficacy of erector spinae plane block in lumbar spine surgery: A systematic review and meta-analysis. J Clin Anesth 2022;78:110647. https://doi.org/10.1016/j.jclinane.2022.110647
- Agarwal P, Pierce J, Welch WC. Cost analysis of spinal versus general anesthesia for lumbar diskectomy and laminectomy spine surgery. World Neurosurg 2016;89:266-71. https://doi.org/10.1016/j.wneu.2016.02.022
- Walcott BP, Khanna A, Yanamadala V, et al. Cost analysis of spinal and general anesthesia for the surgical treatment of lumbar spondylosis. J Clin Neurosc 2015;22:539-43. https://doi.org/10.1016/j.jocn.2014.08.024
- Sun Q, Zhang C, Liu S, et al. Efficacy of erector spinae plane block for postoperative analgesia lumbar surgery: a systematic review and meta-analysis. BMC Anesthesiol 2023;23(1):54. https://doi.org/10.1186/s12871-023-02013-3
- Liu H, Zhu J, Wen J, et al. Ultrasound-guided erector spinae plane block for postoperative short-term outcomes in lumbar spine surgery: a meta-analysis and systematic review. Medicine (Baltimore)2023;102(7):e32981. https://doi.org/10.1097/MD.0000000000032981
- Wetmore DS, Dalal S, Shinn D, et al. Erector spinae plane block reduces immediate postoperative pain and opioid demand after minimally invasive transforaminal lumbar interbody fusion. Spine (Phila Pa 1976)2024;49(1):7-14. https://doi.org/10.1097/BRS.0000000000004581
- De Biase G, Otamendi-Lopez A, Chen S, et al. Impact of postoperative fatigue following minimally-invasive lumbar spine surgery. J Clin Neurosci 2023;112:64-7. https://doi.org/10.1016/j.jocn.2023.04.013