The Safety of "Rescue Blocks"
Cite as: Horazeck C, Gadsden J, Jones JW. The safety of "rescue blocks". ASRA News 2020;45. https://doi.org/10.52211/asra110120.069
Despite a lack of clinical guidance, the common fear of needle trauma to a partially anesthetized nerve or additional local anesthetic administration resulting in local anesthetic systemic toxicity (LAST) requires physicians to consider the risks and benefits associated with supplementing or rescuing a failed nerve block. Nerve block failure may result in significant consequences, including conversion to general anesthesia, escalation of pain, increased opioid exposure, delayed recovery and discharge, unplanned emergency room visits or hospital readmissions, chronic pain, cardiovascular events, and patient dissatisfaction.
By routinely utilizing single injection and continuous peripheral nerve blocks, anesthesiologists are brought into an enviable position of positively influencing both patient-centered and financial outcomes.
The growing awareness that opioid reduction yields significant financial benefit[1] and the new concerns that surgery is a significant risk factor for long-term opioid use and addiction[2-4] have both increased the pressure to reduce undue opioid exposure at the time of surgery. By routinely utilizing single injection and continuous peripheral nerve blocks, anesthesiologists are brought into an enviable position of positively influencing both patient-centered and financial outcomes. At the same time, this privilege may add significant pressure to reliably achieve nerve block success.
With an improved ability to facilitate direct visualization of local anesthetic spread and an enhanced ability to achieve nerve blockade with reduced volumes of local anesthetic (and risk of LAST),[5] ultrasound guidance for regional anesthesia has resulted in a greater degree of reliability and safety than ever before. Many traditional nerve blocks can now be performed without the requirement of the needle contacting the nerve. Further, numerous plane block techniques, often considered safer alternatives,[6-7] as well as more easily identified distal sites (including individual nerves), provide pristine anatomic locations remote to the original nerve block sites. These factors potentially expand the opportunity to safely perform so-called “rescue blocks” when single injections fail or continuous infusions are unexpectedly interrupted (Figure 1).
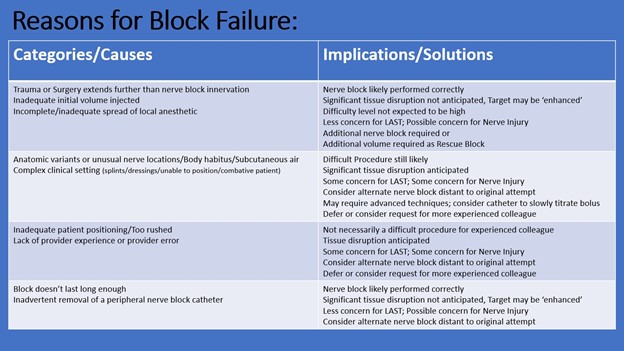
Figure 1: Categories and implications of nerve block failures
Ultrasound is not foolproof, however, and may create misleading optical illusions. In addition, rescue blocks often entail sonoanatomy that has been altered dramatically by tissue plane disruption, injected air from previous attempts, or the surgical procedure. Further, encountering patients in severe pain, half-sedated, who cannot tolerate optimal positioning and are encumbered by new surgical dressings presents challenges even when sonoanatomy is pristine.
While there are risks associated with a rescue block, the risks of opioids or other analgesics are not inconsequential, particularly for some patient populations. But when should one replace a failed nerve block? Which patients might receive the greatest benefit or represent a particularly increased risk from further attempts?
What is a "failed block" anyway?
Before a decision is made whether or not to rescue a block, the clinical effectiveness should first be evaluated, and the degree of failure should be defined. Clinical effectiveness describes the ability of the nerve block to adequately fulfill goals for an individual patient. Clinical effectiveness may occur despite an incomplete distribution or a diminished concentration of local anesthetic at the target site.
The ability of a nerve block to mitigate the consequences of postoperative pain (and/or opioid exposure) can have a much greater impact for certain patient populations, thereby potentially altering the goals of a block.[8-9] The degree and expected timeframe of severe pain, recognized risk factors in individual patients, and recovery goals should be taken into consideration when determining if a nerve block is considered clinically effective.
Degree of nerve block failure is a technical term that will be described here simply as complete or partial. Complete failure indicates that there is no evidence of sympathetic, sensory and/or motor blockade within the desired or expected nerve distribution. Partial failure indicates an objective degree of sympathetic, sensory and/or motor blockade in the desired or expected nerve distribution.
Complete failures typically represent intramuscular injections. The primary concern is that all the local anesthetic has been injected into highly vascularized regions which hastens vascular uptake. The likelihood of LAST with subsequent local anesthetic injections is elevated. Since no local anesthetic is near the target nerves with a complete failure, sensation should be fully intact. With the use of ultrasound guidance in an alert patient, concerns about direct needle trauma to a target nerve may be relatively reduced; however, the efficacy of nerve stimulation may still be unreliable if blood or other fluids are present near the target nerves.
Partial failures likely indicate that the local anesthetic (or at least a portion) was properly injected in the appropriate neurovascular plane adjacent to the intended nerve/nerve bundle, but there was either an inadequate mass of local anesthetic present or a suboptimal spread of the local anesthetic. The reason for partial failure may be twofold. First, local anesthetic was deposited near the nerve (intramuscularly or within other tissues) and in close enough proximity to the nerve to result in significant diffusion to the appropriate target location. Second, it could be due to inadequate spread within the correct location. This may be due to anatomic variation, inadequate opening of fascial planes, or injection of local anesthetic in a location that is incongruent with the entire surgical incision. Injections within the correct tissue plane represent a relatively lower risk of LAST (compared to a complete failure). Tissue distortion after either failure type may be significant, and the introduction of air can make visualization more challenging. However, appropriately injected local anesthetic may actually be helpful due to tissue plane and nerve enhancement. Nerve stimulation is likely less reliable with partial than with complete failures.
Assessing a failed nerve block
Patients often conflate their rated pain level with anxiety or unusual sensations (numbness and tingling is commonly reported as pain). Patients may also be manipulative or unable to respond accurately due to unfamiliarity with nerve blocks and expectation. Identifying the exact location and characterization (sharp/burning vs pressure) of the pain complaint may reveal that a long incision is only felt as pressure or is painful only at one small point.
Physical examination should include an evaluation of motor strength, sympathetic changes, and sensory loss with attention focused on the innervations of individual peripheral nerves. A control site, where cold sensation is expected to be normal, should be presented to the patient to provide a reference point and improve patient report accuracy. Wiping an alcohol pad or ice across or through different nerve innervations will aid the patient in differentiating absence of cold, a relative decrease in cold, and normal cold sensation. Wiping over a clear plastic dressing out of the patient’s field of vision may be helpful in confirming unreliable patient reporting of cold sensations. Objective findings such as inability to ambulate, use of incentive spirometry, and ability to cough are important and should validate considerations for a rescue block.
Prior records may provide important information. Abrupt heart rate and blood pressure increases (or their absence) at incision is highly suggestive of nerve block failure (or success). However, a recent ephedrine dose also could be a confounding factor. Be mindful of these changes when surgical fields have dual nerve innervation such as the ankle. Even in a well-functioning nerve block, tourniquet pain will reliably break through after about one full hour and is represented as a slow rise in heart rate and blood pressure. This should not prompt a rescue block. Giving a further bolus through a nerve block catheter may be considered. Dynamic ultrasound scanning with Doppler and observed injection of normal saline, air, or local anesthetic through a peripheral nerve catheter provides helpful information.
 Figure 2: A framework to systematically evaluate the decision to perform a rescue block
Figure 2: A framework to systematically evaluate the decision to perform a rescue block
Conclusions
The utilization of regional anesthesia has changed dramatically in a very short time period. With the introduction of ultrasound guidance and other advancements in the field, new techniques and options have been discovered. Nerve blocks are performed today with greater precision, reliability, and safety for an ever-expanding range of applications. The significance of many of the positive outcomes associated with regional anesthesia has been highlighted in the setting of the current opioid crisis and increased efforts to reduce healthcare costs.
The question that must be faced by novices and experts alike is whether or not we are in a position to challenge the dogma and previously held assessments of risk assessments that dissuade the consideration of replacing a “failed” nerve block. Has the balance of risks and benefits now shifted toward performing rescue blocks routinely? Are there today some circumstances for particular patients in unique situations that should push us to change our mindset about the relative risks of this practice? Should we write our informed consent forms differently? Do we have ways of accurately quantifying those risks and benefits?
The depth of these questions is far beyond the scope of this discussion, and until further guidelines are published, anesthesiologists must consider their own level of expertise and use their own clinical judgement when caring for individual patients (Figure 2). There are a number of strategies and clinical pearls, however, that can guide the anesthesiologist facing this difficult situation.
Clinical Pearls
- Get the facts! Do not rely solely on patient report of pain. Use available objective signs of clinical success such as stable vital signs at surgical incision or post-operative physical exam. You may not even need to perform a rescue block. A physical exam may reveal that a saphenous nerve block to supplement a successful sciatic nerve block is indicated.
- Why did the nerve block fail? Know the clinical scenario before considering a second attempt. Previously encountered imaging difficulties might be easily overcome if patient positioning can now be improved. The same may not be true if significant subcutaneous air was the cause imaging difficulties. Scanning to reevaluate the sonoanatomy may be a very helpful exercise in either scenario.
- Think outside the box! Consider alternate locations that may provide the same outcome: transversus abdominis plane vs rectus sheath vs quadratus lumborum vs erector spinae plane. Consider an infraclavicular approach if a supraclavicular approach was previously performed, or consider blocking individual nerves in the forearm.
- Next time… Update procedure notes to provide specific, helpful information (similar to difficult airway notes that will guide anesthesiologists in the future as to whether someone with more experience should be contacted up front in the case of very difficult cases should they return for a similar procedure) (Figure 3).
- Fentanyl, not morphine, if required for analgesia. In the meantime, short-acting opioids will minimize sedation and respiratory depression after the rescue block is successfully performed.
- Got help? Rescue blocks may be performed after hours or in remote locations. Make sure monitoring and adequate assistance is available before proceeding.

Figure 3: Procedure notes can provide helpful information to guide future decision-making.
References
- ^ Oderda GM, Gan TJ, Johnson BH, Robinson SB. Effect of opioid-related adverse drug events in selected surgical patients. J Pain Palliat Care Pharmacother. 2013;27(1): 62-70.
- ^ Alam A, Gomes T, Zheng H, Mamdani MM, Juurlink DN, Bell CH. Long-term analgesic use after low-risk surgery: a retrospective cohort study. Arch Intern Med. 2012;172 (5):425-30.
- ^ Sun EC, Darnall BD, Baker LC, Mackey S. Incidence of and risk factors for chronic opioid use among opioid-naive patients in the postoperative period. JAMA Intern Med. 2016;176:1286-93.
- ^ Brummett CM, Waljee JF, Goesling J, et al. New persistent opioid use after minor and major surgical procedures in US adults. JAMA Surg.2017;152(6):e170504.
- ^ Barrington MJ, Kluger R. Ultrasound guidance reduces the risk of local anesthetic systemic toxicity following peripheral nerve blockade. Reg Anesth Pain Med. 2013;38(4):289-99.
- ^ Forero M, Adhikary SD, Lopez H, Tsui C, Chin KJ. The erector spinae plane block: a novel analgesic technique in thoracic neuropathic pain. Reg Anesth Pain Med. 2016;41(5):621-7.
- ^ Voscopoulos JC et al. The ultrasound-guided retrolaminar block. Can J Anaesth. 2013;60:888–95.
- ^ Chou R, Gordon DB, de Leon-Casasola OA, et al. Management of postoperative pain: a clinical practice guideline from the American Pain Society, the American Society of Regional Anesthesia and Pain Medicine, and the American Society of Anesthesiologists' Committee on Regional Anesthesia, Executive Committee, and Administrative Council. J Pain. 2016;17(2):131-57.
- ^ The Joint Commission. Pain assessment and management standards for hospitals. August 29, 2017. Available at: https://www.jointcommission.org/-/media/tjc/documents/resources/patient-safety-topics/sentinel-event/r3_report_issue_11_pain_assessment_8_25_17_final.pdf. Accessed October 21, 2020.
Leave a commentOrder by
Newest on top Oldest on top