How I Do It: Kyphoplasty
Cite as: Malik T. How I do it: kyphoplasty. ASRA News 2020;45. https://doi.org/10.52211/asra110120.070
Osteoporosis is the most common bone metabolic disease. It increases in frequency with age and is the most common reason for developing a vertebral compression fracture.[1] The National Osteoporosis Foundation estimates 10 million people in the United States over age 50 suffer from osteoporosis and an additional 44 million have osteopenia.[2] By age 50, the remaining lifetime fracture risk is 1 in 2 for women and 1 in 5 for men.[3] The disease is responsible for 2 million fractures a year at great economic cost in the United States.[4] Annually, 60,000 office visits and 70,000 admissions occur from osteoporotic vertebral compression fractures (VCF) in the United States.[5] There is no cure for osteoporosis, and, with an aging population, the problem is bound to worsen. It is projected that by 2025, 3 million osteoporotic fractures will cost $25 billion a year in related health care costs.[6] Currently, a vertebral compression fracture is managed either conservatively or with vertebral augmentation therapy.
Some osteoporotic VCFs result in minimal or mild pain. Symptoms typically subside in 6–8 weeks as healing occurs. Conservative or medical management usually involves pain management with analgesics or limiting activities/bed rest, back braces, and physical therapy with the aim to keep the patient functional and comfortable. Patients with disabling, severe pain require hospitalization. In any situation, either at home or in the hospital, a significant decrease in ambulation has negative consequences. A bone mass loss can be seen within 2 days of bed rest;[7] one week of bed rest results in a rate of bone loss that is 50 times the normal age-related rate.[8] Two weeks of bed rest results in 15% of aerobic capacity and lower extremity strength loss, equivalent to 10 years of age-related loss.[9] The use of narcotics to improve ambulation is not very effective and is associated with the adverse effects of sedation, nausea, and constipation that further deteriorate physical deconditioning. Half of hospitalized patients need ongoing care after discharge,[5] and chronic pain develops in 40%.[10] Therefore, although medical management is widely used, there are significant negative effects.
Pathophysiology of Pain
Most osteoporotic vertebral compression fractures are stable and do not cause any nerve compression or threaten the spinal cord and therefore can be managed conservatively. Pain from compression fracture is multifactorial. In the acute phase (first 4-6 weeks), there is swelling and inflammation within the vertebral body, vertebral body cortical wall, and/or the endplate deformities. In addition, there is altered axial load transmission along the anterior column (along the vertebral bodies) and the posterior column (facet joints). Pain at rest may be bearable, but dynamic pain/pain with movement is invariably worse as compressive forces produced during movement result in micro-motion at the fracture level. In supine or prone positions, the axial load decreases and the pain subsides. The main purpose of vertebral augmentation is to quickly restore functionality of the patient by alleviating pain, especially with movement, by stabilizing the fracture using polymethylmethacrylate (PMMA), which glues together broken pieces, thereby improving the mechanics of the spine by restoring vertebral body (VB) height. PMMA polymerizes on deposition; this is an exothermic reaction with the temperature rising above 90oC. This causes the neurolysis of nerves that helps with pain control as well as killing nearby malignant cells.
Indication
Kyphoplasty is indicated for the treatment of disabling pain from a vertebral body compression fracture. Treating pain that is not from a VCF or pain not limiting ambulation will result in a suboptimal outcome. The best outcome is achieved when patients are carefully selected. The patient should fulfill three criteria:
- Presence of acute/subacute compression vertebral body fracture
- Mechanical back pain from the fracture level limiting ambulation
- Failed initial conservative treatment
Failure of conservative therapy is defined as failure in pain improvement within a few days despite the use of potent analgesics, failure in significant pain improvement within a few weeks with conservative therapy, or inability of the patient to tolerate analgesics to improve functional status. Improvement in the functional status and not the visual analog score is the key point when deciding if conservative therapy is succeeding or failing.
Other than VCF, kyphoplasty procedures also are performed for strengthening a VB weakened by malignancy or in symptomatic VB microfractures without loss of height.
Patient Evaluation
A thorough patient history focuses on:
- Evaluating pain (back pain vs radicular pain)
- Uncovering predisposing factors (osteoporosis, malignancy, trauma) in an effort to reduce the risk of subsequent fractures
- Determining the duration of painful symptoms (acute vs chronic)
- Determining functional limitations imposed by pain (critical for determining urgency of interventions)
- Identifying the various conservative modalities tried so far.
The physical exam is focused on eliciting midline back tenderness over the fractured vertebra and ruling out any neurologic deficits.
Blood work is not generally indicated unless it is part of a broader medical workup. Tests to exclude coagulopathy are also not needed unless indicated based on medical history.
Imaging is the most important component of the evaluation. Plain spine x-ray may point to a fractured vertebra but cannot establish timing or morphology of the fractured bone. Magnetic resonance imaging (MRI) with short tau inversion recovery (STIR) images is very useful in evaluating the acuteness of the fracture, anatomy of the fracture (extent of collapse, end plate deformity, retroplusion of any bony fragment), evaluating defects in the cortical wall especially posterior wall, presence of a malignancy and spread beyond the body wall/epidural space involvement, and any evidence of nerve compression or spinal cord compression. MRI with STIR images best evaluates edema or inflammation within the VB by suppressing fat signals (Figure 1). Persistent edema in the VB, as visualized on MRI STIR images, is a marker that the fracture has not completely healed and may still be a cause of pain and amenable to vertebral augmentation therapy.[11] If needed, spine computerized tomography (CT) is conducted to evaluate cracks in the VB wall. This information may be more critical if there is questionable integrity of the posterior vertebral wall demonstrated on MRI. Similarly, the pedicle size and integrity can be reasonably well visualized on CT images. If an MRI study is contraindicated, the spine should be evaluated with single-photon emission computerized tomography scan or positron emission tomography scan. Each study is equally effective in evaluating the anatomy of VB for fracture or deformity and for the presence of abnormal blood flow within the VB, which correlates with an unhealed fracture.[12,13] Radiological images should be reviewed to map out a trajectory plan for the procedure. It’s important to measure size of the pedicles (diameter in mm), trajectory lines of collapsed endplates, integrity of posterior VB wall, and shape of the VB in transverse plane. Many misadventures can be prevented if proper dimensions of the collapsed VB are properly studied before the procedure. I use these dimensions to plan for cannula trajectory, opt for unipedicular vs bipedicular approach, and identify which parts of the VB to target or avoid. Studying the images also can help one decide if a transpedicluar approach is feasible or an extrapedicular approach is needed.
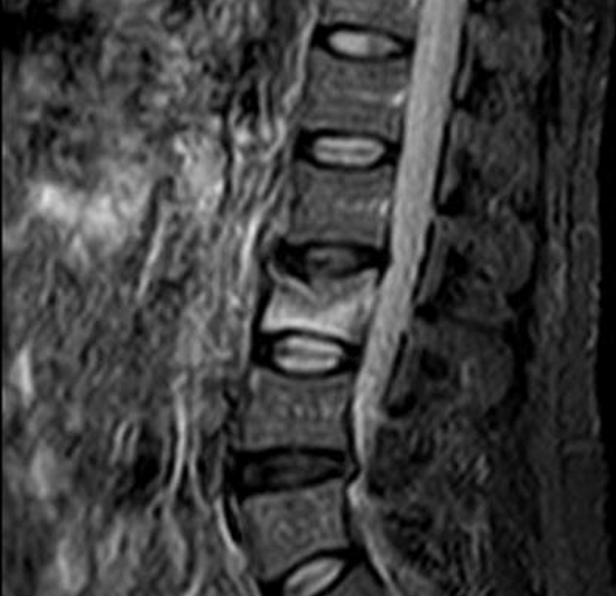
Figure 1: MRI with T2 short tau inversion recovery (STIR) imaging (vertebral body is bright)
Contraindication: Most contraindications to kyphoplasty are relative. However, invasive intervention in the presence of the following conditions deserves caution, and these patients may be better served with more conservative therapy:
- Systemic infection
- Healed fracture/old fracture/non-painful lesion
- Discitis or osteomyelitis at the level of planned intervention
- Coagulopathy (INR > 1.2, platelets less than 100k)
- Loss of posterior body wall integrity
- Retroplusion especially if causing stenosis
- Pars plana (more than 90% collapse of the VB)
- Tumor invading the epidural space
A crack in the posterior wall of the body or retroplusion of the wall is not an absolute contraindication. If the retropulsed part is not free floating and the posterior longitudinal ligament is intact, the procedure can still be performed.[14] If the posterior wall has a crack, cement can be injected very anteriorly in the VB, but do so carefully so it does not leak back into the epidural space.
Technique
Anesthesia: Most kyphoplasty procedures generally can be performed with mild to moderate sedation. However, the procedure also can be performed with local or general anesthesia.[15] Cases also have been done using epidural anesthesia.[16] The key is to have an immobile patient during the procedure as any patient movement will require an adjustment of the x-ray machine, resulting in frequent interruptions.
Position: The patient is positioned prone with their arms secured so that they don’t interfere with the lateral view of the spine. The operating room table should have enough clearance for free movement of the C–arm between anterior-posterior (AP) and lateral positions. A Jackson spine table with a Wilson frame is quite useful as it relieves pressure of the abdomen and improves respiratory dynamics in the patient. Positioning can help restore the height of a VB; exaggerating lordosis in the lumbar spine or minimizing kyphosis in the thoracic area will help restore VB height.
Area Preparation: The skin over the fractured VB is outlined with sterile drapes. The skin is cleaned with 3% chlorhexidine in 70% isopropyl alcohol or an iodine-based solution. Full surgical drapes are used for this procedure. The target area is covered with an antimicrobial drape (eg, Ioban®). Patients are given IV antibiotics (eg, cefazolin) before the incision.
Imaging: Unless CT or an O-arm guidance are being used (which provide real time 3-D images of the anatomy during the procedure) the procedure is most commonly performed using fluoroscopic guidance. Two C-arms are very helpful if they can be properly placed around the patient. Otherwise, one will suffice.
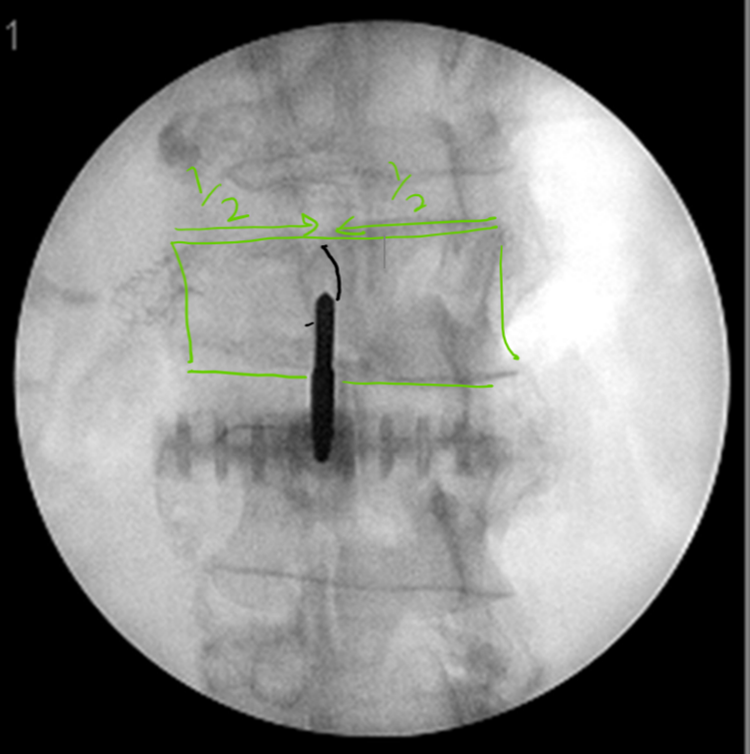
Procedure: Prior to initiating the procedure, it is important to refresh your understanding of the anatomy of the collapsed VB by viewing existing radiological images. It is important to recall the severity of end-plate collapse – especially in the middle – and the thickness and trajectory of the pedicle as it attaches to the posterior aspect of the VB. This pedicular trajectory is important when using a transpedicular approach and achieves greater significance at the thoracic level. With one C-arm, the enface view is obtained and the cannula is placed via the pedicle using a bulls-eye view, with the aim to perform vertebral augmentation using a unipedicluar approach. En face view is similar to obtaining a scotty dog view for facet joint procedures (Figure 2). In AP view, the C-arm is adjusted craniocaudally till the superior end plate is aligned. This will bring the pedicle in the upper third of the VB or in the middle if endplates are too collapsed. Then oblique the C-arm till the pedicle medial border is clearly seen and is the sharpest. This angle can be measured on the MRI or CT images using imaging software (Figure 3). If pedicle anatomy is distorted by the disease, then traditional AP and lateral imaging views should be used. Once a proper view has been obtained, it’s good to note the degree of C-arm tilt and its obliqueness so that the same view can be obtained again quickly, in case the C-arm has to be moved.
Figure 2: Pedicle should be placed roughly the of the vertebral body for unipedicluar en face approach
| 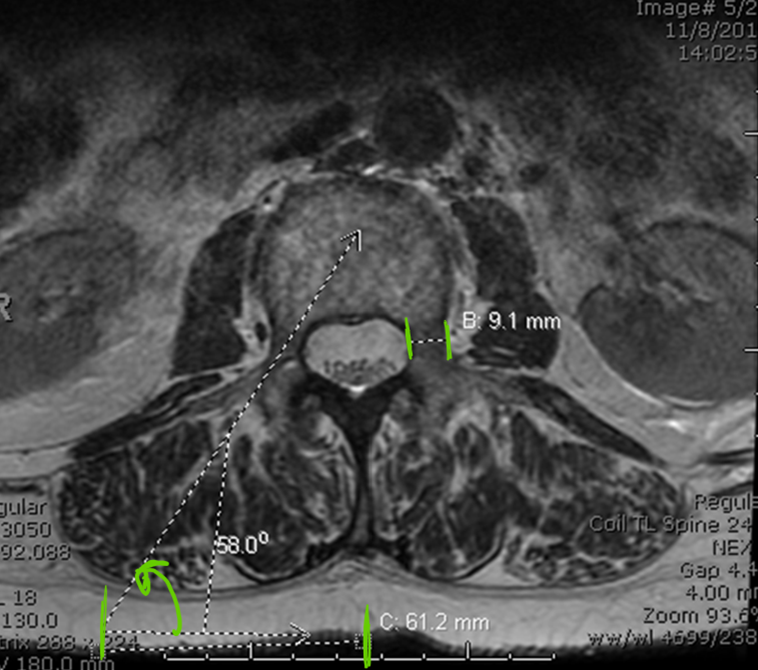
Figure 3: Pedicle size, skin entry point, C-arm oblique angle can be all be measured before starting the procedure
|
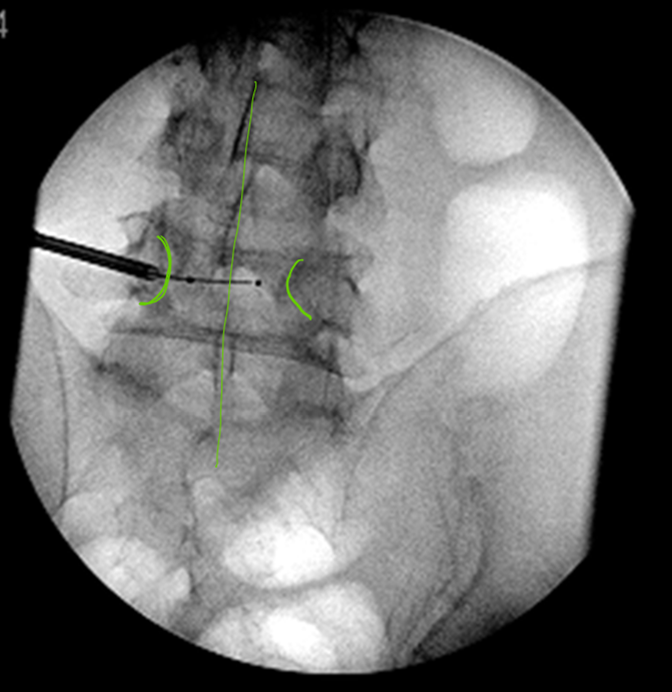
Local anesthetic should be liberally used even if the case is performed under general anesthesia. It’s helpful to anesthetize the tract up to the periosteum using an 18 g or 22 g spinal needle. The spinal needle, once in place, can be used to confirm the appropriateness of the trajectory in AP/lateral view before using the 11 g introducer needle. The working cannula can be loaded with either a diamond tip or bevel tip stylet; the bevel tip provides some directional control. The cannula is advanced into the cavity of the VB using a bullseye view. The key point is to not violate the medial border of the pedicle until the needle tip enters the VB. Lateral and AP views should be used to confirm cannula position once half-way through the pedicle and again right before entering the body. The cannula should be advanced until it’s within the posterior fourth of the VB. When using unipedicular approach, the tip of the cannula should be at the junction of middle and lateral third of the VB in AP view. The bone drill is then inserted via the cannula to create a space for the introduction of the balloon. The drill should be advanced in lateral view, until it’s just short of the anterior VB wall; at this point the tip of the drill should be close to the pedicle of the opposite side in AP view. The balloon, filled with radio-opaque agent, is inflated slowly to allow for careful monitoring of the pressure gauge and the volume injected into the balloon (Figure 4). This is done under flouro guidance. Once the balloon is inflated to an appropriate volume or results in abutting the end plates or the side walls of the VB, further inflation is aborted. The balloon remains inflated and cement is mixed for injection. If more than one level requires treatment, then those additional levels are accessed before cement is mixed. Once cement is mixed and ready to be used, it is loaded into the bone filler, each of which holds 1.5 ml cement. Nowadays, a delivery system is available which can be used for cement injection that negates the need to load the bone filler again and again. Once the optimal consistency is achieved (like toothpaste, does not drip), the balloon is deflated and removed. The bone filer is inserted all the way into the VB just short of the anterior wall; cement is injected slowly, under live fluoroscopic guidance. Intermittent AP view but frequent lateral views are important to ensure safe and even spread of the cement within the VB. The process is repeated until the optimal amount and spread of cement has been achieved. If the cement leaks back toward the posterior aspect of the VB, the injection is stopped. If adequate cement spread has been achieved, no additional cement is injected in the VB. If the initial cement spread is inadequate, 2-3 minutes should be allowed to elapse to allow for the leaked cement to harden. At this point, cautious cement injection can again proceed. If the injected cement no longer leaks back, then the cement injection may continue. Optimally, spread should be even in all quadrants of the VB and fluffy looking, interdigitating with the native cancellous bone (Figure 5). If the bone fragments or clefts are present as seen on MRI, those should be filled and interconnected.
Figure 4: Balloon is across midline with unipedicluar approach |
Figure 5: Evenly distributed cement |
The cannula is left in place until the cement has hardened. This is confirmed by tapping the cement with a stylet of the bone filler; it should sound like tapping a brick. The cannula is then removed under flouro guidance ensuring the cement is not tracking back.
A final AP and lateral image of the VB without any hardware is obtained. The skin incision is closed with a 3-0 vicryl or monocryl suture and incision covered with skin glue (eg, Dermabond®). The patient is taken to the post-anesthesia care unit. Neuro intactness is documented in the recovery room. The patient is discharged once they have recovered from the effects of anesthesia. Carefully considering a few clinical pearls in Figure 6 will maximize the opportunities for success with this procedure.
- Proper patient selection ( Back pain < 3 months, localized tenderness, MRI with STIR images(+)
- Focus on functional improvement not VAS
- Know your anatomy – use CT scan to delineate VB anatomy if needed.
- Measure VB and pedicle dimensions and angle to plan for needle trajectory
- Take time to get optimal fluoroscopic views (aim for perfect lateral, AP or oblique view)
- Do not guess when reading fluoroscopy images during the procedure.
Figure 6. Clinical pearls
Variations on the technique: Variations to the core technique of balloon kyphoplasty have been described. These include radiofrequency ablation used to treat a tumor in the VB (OsteoCool RF®), depositing a coil implant and filling it with cement thus lowering cement leakage risk (Kiva system®), and depositing metal implants inside the VB, which are then screwed open to lift the depressed endplates, followed by cement injection inside the implant, which also lowers the risk of cement leakage (SpineJack® System, Stryker).
Post-procedure instructions: Patients are instructed to ambulate as tolerated once at home. Procedural pain should abate in a few days, and pain clinic follow-up should occur 2 weeks following the procedure.
Question of cement volume injection and distribution: The VB bone strength is restored with just 2-3 ml of cement. There is a suggestion that 15%-30% of VB volume filling with PMMA will restore VB strength and stiffness. This translates into 4 ml cement for a thoracic vertebra and 6 ml for a lumbar vertebra. The distribution should be even and from one end plate to another. Uneven or lopsided cement filling will distort load transmission through the VB. However, it is important to maximize the volume of cement administered while ensuring that a safe filling volume is administered.[17-20]
Complications: The procedure is not immune from complications (Figure 7). Fortunately, these complications are relatively uncommon.[21] The most common complication tends to be less than optimal pain relief. This is due to poor patient selection (treating an old fracture). More serious complications include damage to surrounding structures resulting in pneumothorax, rib fracture, or hematoma. There is a chance that adjacent level vertebra may develop a new compression fracture. The most feared complication is cement leakage into the spinal canal causing spinal cord or nerve damage. Intravascular leakage and embolization of cement can cause cardiopulmonary compromise and even death. Infection is an incredibly rare but devastating event that would require corpectomy and aggressive antibiotic treatment.
- Infection (superficial /deep)
- Bleeding
- Rib fracture
- Pneumothorax
- Cement leakage into the discs, surrounding tissues
- Cord compression from acute spinal stenosis from cement leak
- Nerve root damage
- Intraoperative hypotension (cement/fat embolism
- Pulmonary Embolism
- Cardiac arrest
Figure 7. Complications
Effectiveness
Jensen et al introduced vertebroplasty to the United States in 1993. Her teams’ findings were first published in the American Journal of Neuroradiology in 1997.[22] In 2007, positive preliminary data led medical societies to endorse vertebral augmentation as safe and effective for painful osteoporotic VCFs refractory to medical management. The procedure has been studied extensively (Table 1).
Table 1: Kyphoplasty clinical trials
| Trials | Type of Trial | Primary End Points | Outcome |
| FREE Trial (2009)23 | Randomized, controlled (kyphoplasty vs Medical Management) | SF-36 at 1 month vs baseline | SF-36 improved by 5.2 points at 1 month |
| Kallmes (2009)24 | Randomized double-blind (vertebra vs sham) | Roland-Morris Disability Questionnaire and VAS at baseline vs 1 month | No difference between the two groups |
| Buchbinder (2009)25 | Randomized, double-blind, placebo-controlled | VAS at 3 months | No difference between the groups |
| VERTOS II (2010)26 | Open label randomized, vertebroplasty vs medical mgt. | VAS at 1 month and 1 year | VAS improved by 2.6 at 1 month and by 2.0 at 1 year |
| VAPOUR (2016)27 | Randomized, double-blind, placebo-controlled trial | VAS at <4/10 at 14 days | 44% vs 21% had <4/10 VAS at 14 days |
| VERTOS IV (2018)28 | Randomized, double blind, sham controlled clinical trial. | VAS at 1 week, and at 1,3,6,12 months | No difference between the two groups all along |
Conflicting data has resulted in the withdrawal of public funding for the procedure in some locations (eg, Australia in 2011). However, the procedure remains available to patients in the United Kingdom if they have failed conservative therapy. The Centers for Medicare and Medicaid Services recently developed new payment guidelines to guide reimbursement for vertebral augmentation therapy. The guidelines considered the lack of conclusive evidence when treating compression fractures of different etiologies (Figure 8).
PVA (percutaneous VERTEBROPLASTY (PVP) or kyphoplasty (PKP)) is covered in patients with BOTH the following:
- Inclusion criteria (ALL are required):
- Acute (< 6 weeks) osteoporotic VCF (T5 – L5) by recent (within 30 days) advanced imaging (bone marrow edema on MRI or bone-scan/SPECT/CT uptake)
- Symptomatic (ONE):
- Hospitalized with severe pain (Numeric Rating Scale (NRS) or Visual Analog Scale (VAS) pain score ≥ 8)
- Non-hospitalized with moderate to severe pain (NRS or VAS ≥5) despite optimal non-surgical management (NSM) (ONE):
- Worsening pain
- Stable to improved pain (but NRS or VAS still ≥5) (with ≥ 2 of the following):
- Progression of vertebral body height loss
- > 25% vertebral body height reduction
- Kyphotic deformity
- Severe impact of VCF on daily functioning (Roland Morris Disability Questionnaire (RDQ) >17)
- Multidisciplinary team consensus (2) (ALL are required):
- Referring physician (e.g., rheumatologist, endocrinologist)
- Treating physician (i.e., performing the PVA)
- Radiologist
- Neurologist
Figure 8. CMS Coverage Guidance 2020
Conclusion
Kyphoplasty is a safe and effective therapy in select patients with acute and painful compression fractures.
References
- ^ Rachner T, Khosla S, Hofbauer L. Osteoporosis: now and the future. 2011;377:1276–87.
- ^ NOF Releases Updated Data and National Breakdown of Adults Age 50 and Older Affected by Osteoporosis and Low Bone Mass. Available at: https://www.nof.org. Accessed on March 20, 2020.
- ^ Johnell O, Kanis J. Epidemiology of osteoporotic fractures. Osteoporos Int. 2005;16(suppl 2):S3–7.
- ^ Singer A, Exuzides A, Spangler L, et al. Burden of illness for osteoporotic fractures compared with other serious diseases among postmenopausal women in the United States. Mayo Clin Proc.2015;90:53–62.
- ^ a b Gehlbach SH, Burge RT, Puleo E, Klar J. Hospital care of osteoporosis-related vertebral fractures. Osteoporos Int. 2003;14:53–60.
- ^ Burge R, Dawson-Hughes B, Solomon DH, et al. Incidence and economic burden of osteoporosis-related fractures in the United States, 2005–2025. J Bone Miner Res. 2007;22:465–75.
- ^ Baecker N, Tomic A, Mika C, et al. Bone resorption is induced on the second day of bed rest: results of a controlled cross over trial. J Appl Physiol.2003;95:977–8.
- ^ Marie PJ, Kassem M. Extrinsic mechanisms involved in age-related defective bone formation. J Clin Endocrinol Metab. 2011;96:600–9.
- ^ Kortebein P, Symons TB, Ferrando A, et al. Functional impact of 10 days of bed-rest in healthy older adults. J Gerontol A Biol Sci Med Sci.2008;63:1076–81.
- ^ Venmans A, Klazen CA, Lohle PNM, Mali WP, van Rooij WJ. Natural history of pain in patients with conservatively treated osteoporotic vertebral compression fractures: results from VERTOSII. AJNRAm J Neuroradiol. 2012;33:519–21.
- ^ Park SY, Lee SH, Suh SW, Park JH, Kim TG. Usefulness of MRI in determining the appropriate level of cement augmentation for acute osteoporotic vertebral compression fractures. J Spinal Disord Tech. 2013;26(3):E80–5.
- Yong-Bo L, Zhengi X, Wang R, et al. SPECT-CT versus MRI in localizing active lesions in patients with osteoporotic vertebral compression fractures. Nucl Med Commun. 2018;39(7):610–7.
- Schmitz A, Risse JH, Textor J, et al. FDG-PET findings of vertebral compression fractures in osteoporosis: preliminary results. Osteoporos Int. 2002;13(9):755–61.
- ^ Venier A, Roccatagliata L, Isalberti M, et al. Armed kyphoplasty: an indirect central canal decompression technique in burst fractures. AJNR Am J Neuroradiol. 2019;40(11):1965–72.
- ^ Liu J, Wang L, Chai M, Kang J, Wang J, Zhang Y. Analysis of anesthesia methods in percutaneous kyphoplasty for treatment of vertebral compression fractures. J Healthc Eng. 2020;2020:3965961.
- ^ Apan A, Cuvas Apan Ö, Köse EA. Segmental epidural anesthesia for percutaneous kyphoplasty: comparison with general anesthesia. Turk J Med Sci.2016;46(6):1801–7.
- ^ Belkoff SM, Mathis JM, Jasper LE, Deramond H. The biomechanics of vertebroplasty: the effect of cement volume on mechanical behavior. Spine (Phila Pa 1976) 2001;26(14):1537–41.
- ^ Liebschner MA, Rosenberg WS, Keaveny TM. Effects of bone cement volume and distribution on vertebral stiffness after vertebroplasty. Spine (Phila Pa 1976) 2001;26(14):1537–41.
- ^ Kaufmann TJ, Trout AT, Kallmes DF. The effects of cement volume on clinical outcomes of percutaneous vertebroplasty. Am J Neuroradio. 2006;27(9):1933–7.
- ^ Kim DJ, Kim TW, Park KH, Moon PC, Kim JO. The proper volume and distribution of cement augmentation on percutaneous vertebroplasty. J Korean Neurosurg Soc. 2010;48(2):125–8.
- ^ Robinson Y, Tschöke SK, Stahel PF, Kayser R, Heyde CE. Complications and safety aspects of kyphoplasty for osteoporotic vertebral fractures: a prospective follow-up study in 102 consecutive patients. Patient Saf Surg. 2008;2:2.
- ^ Yang EZ, Xu JG, Huang GZ, et al. Percutaneous vertebroplasty versus conservative treatment in aged patients with acute osteoporotic vertebral compression fractures: a prospective randomized controlled clinical study. Spine (Phila Pa 1976).2016;41:653–60.
- Chen D, An ZQ, Song S, Tang J-F, Qin H. Percutaneous vertebroplasty compared with conservative treatment in patients with chronic painful osteoporotic spinal fractures. J Clin Neurosci.2014;21:473–7.
Leave a commentOrder by
Newest on top Oldest on top