How I Do It: Managing an “Inherited” Intrathecal Pump Patient
This article originally appeared in ASRA News,Volume 14, Number 2, pp 15-16,20 (May 2014).
Lynn R. Kohan, MD, MS
Assistant Professor of Anesthesiology and Pain Medicine
Fellowship Director and Director of Education,
University of Virginia Pain Management Center
Department of Anesthesiology, University of Virginia
Charlottesville, Virginia
Section Editor: Kevin Vorenkamp, MD
Intrathecal (IT) drug infusion systems are widely used in the treatment of refractory chronic pain conditions, malignant pain, and spasticity.[1,2] In fact, the use of such systems has increased dramatically over the past several years.[3] Appropriate patient selection is paramount to successful treatment outcomes; however patient selection and treatment paradigms vary widely between practitioners. This variability leads to difficulties when “inheriting” a patient with an IT delivery device from another practice.
When evaluating a “pump patient” in this situation, start by assessing the patient’s underlying pain condition, co-morbidities, psychological disposition, and social factors in order to tailor care in an appropriate manner (Figure 1).[4] Make sure IT therapy is being utilized for an indicated condition such as neuropathic pain, failed back surgery syndrome, complex regional pain syndrome, and other disorders that are caused by injury to the central nervous syndrome.[4] In addition, one should also analyze the anatomic location of the catheter tip in the intrathecal canal, as well as the medications utilized, as both of these factors may affect the efficacy of analgesia.[5]
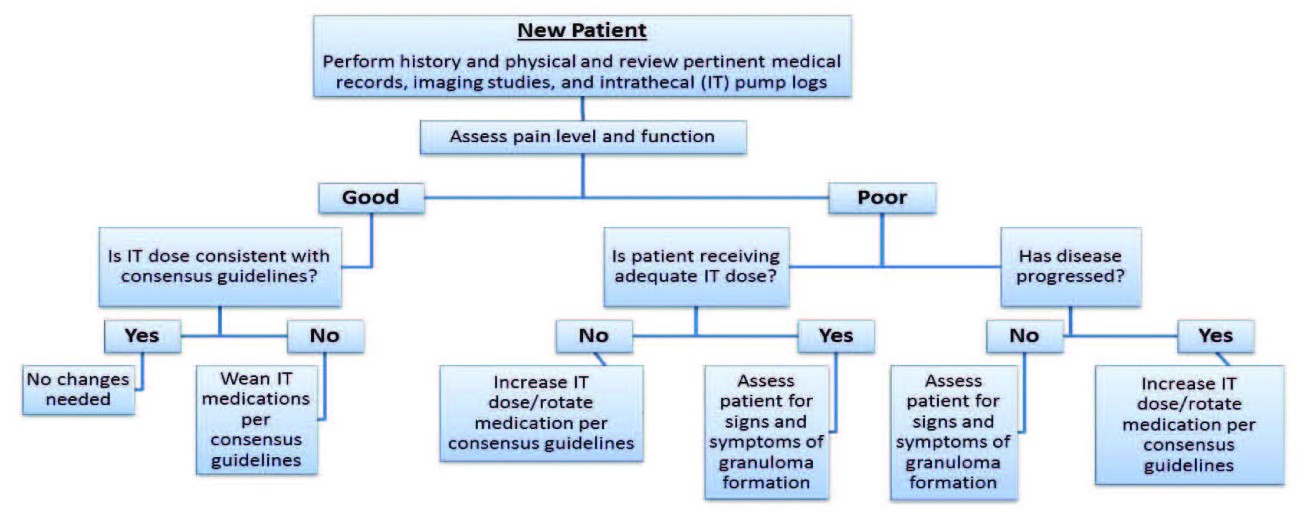
Figure 1. Flow chart to aid clinicians in assessing an “inherited” pump patient according to Polyanalgesic Consensus Conference recommendations.[10]
It is also important to review the patient’s co-morbid conditions as these can have serious impact on adverse effects of the therapy. Conditions such as obstructive sleep apnea (OSA) and chronic lung disease can put a patient at higher risk of respiratory depression. Age factors into dosing recommendations as well. Younger patients usually escalate dosage and develop tolerance more rapidly than older patients.[6,7] Patients with implanted IT devices should continue to be monitored and treated for psychological co-morbidities. It is also important to evaluate the social factors of the patient; the patient must be able to attend clinic appointments and not be impeded by geographical limitations or other issues. Deer and colleagues assert that no co-morbid conditions are absolute contraindications for IT pump therapy as long as patients are properly monitored and dosed cautiously.[8] Therefore, it is essential when assuming care of an existing pump patient that these concomitant conditions be reviewed to ensure that current treatment is appropriate.
Further, one should assess any new patient for adverse effects and complications that can occur from IT delivery systems. Appropriate dosing of medications is essential as IT therapy has been known to cause serious adverse effects, including sedation, respiratory depression, hypotension, opioid-induced hyperalgesia, hypogonadotrophic hypogonadism, and even mortality.[9] In addition, dose and concentration of medication can contribute to the development of granulomas.[9]
When evaluating the patient’s current IT medications and doses, attempts should be made to abide by the 2012 Polyanalgesic Consensus Conference recommendations for both neuropathic and nociceptive pain (Table 1). The lowest dose that is able to achieve analgesia and increased function should be utilized. This may necessitate weaning patients who have been on doses that exceed maximum recommended doses or concentrations; the rationale for doing so must be clearly explained as patients can be expected to exhibit resistance to change.
Table 1. Suggested concentrations and doses for intrathecal agents by the Polyanalgesic Consensus Conference panelists in 2012.[10]
Drug | Maximum Concentration | Maximum dose per day |
Morphine | 20mg/ml | 15mg |
Hydromorphone | 15mg/ml | 10mg |
Fentanyl | 10 mcq/ml | No known upper limit |
Sufentanil | 5mg/ml | No known upper limit |
Bupivacaine | 30mcq/ml | 10mg |
Clonidine | 1000mcq/ml | 40-600mcq/day |
Ziconitide | 100mcq/ml | 19.2 mcq/day |
For the pump patient who does not report improved pain scores and increased function or improved quality of life despite what is deemed adequate dosing, change in the infused drug may be considered. However, the integrity of the pump system including the catheter should first be evaluated, and any new disease process causing worsening of pain should be excluded.[10] One may consider adding an adjuvant prior to changing medications if the patient is tolerating the primary medication without adverse effects.[10] If unable to meet objective goals of treatment, it may be necessary to wean IT medications.
Another important consideration is granuloma formation in patients with worsening pain despite increased dosage of IT medications (Figure 2). Signs and symptoms include decreased response to medications, increased pain, new thoracic spine pain, gastrointestinal pain, lower extremity weakness or paralysis, new bowel or bladder dysfunction, new radicular pain near the dermatomal level of the catheter tip, and any new neurologic sign.[11] Patients who have received high concentrations of IT opioids at any time, those receiving high concentrations with low flow rates, patients with steadily increasing concentrations of opioid medications over time, patients with a history of previous granuloma, and those with disease states such as spinal stenosis or traumatic spinal cord injury are all at higher risk of granuloma formation.[11]
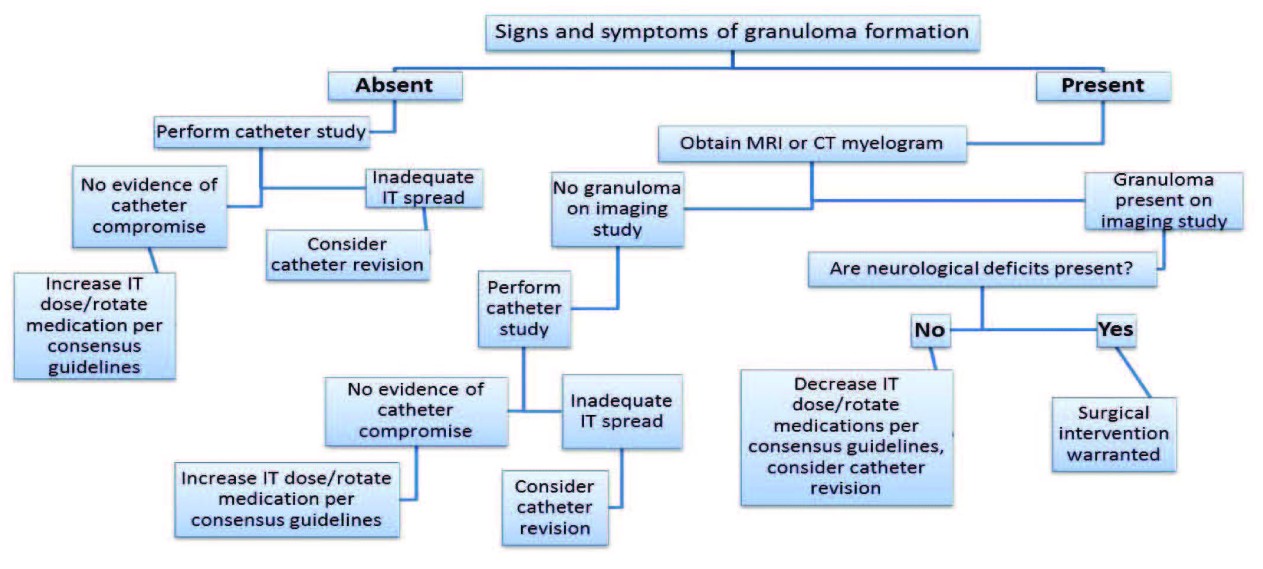
Figure 2. Flow chart to aid clinicians in assessing patients with intrathecal pumps for signs and symptoms of granuloma formation according to Polyanalgesic Consensus Conference recommendations.[11]
To minimize the risk of granuloma formation, pain physicians should use the lowest effective dose and concentration of IT opioid medication and possibly even use bolus dosing instead of a continuous infusion. Other suggestions to minimize the risk of granuloma formation include placing the catheter tip below the level of the conus when possible and using adjuvant non-opioid analgesics or ziconitide.[11] One should also practice vigilant monitoring of patients for early detection of granuloma formation. This should include taking a complete history and physical on all patients receiving IT therapy at least every 6 months and monitoring for signs and symptoms of granuloma formation (Figure 2).
In conclusion, there are many challenges when taking over the care of a patient with an IT drug delivery system. With no clear standards to aid clinicians, it is best to re-evaluate the patient and tailor care to match available recommendations established by the recent Polyanalgesic Consensus Conference. It is strongly recommended that transfers of care involving a pump patient be planned in advance with all parties involved to optimize the overall treatment of the patient. When the patient and new managing physician engage in shared decision making within the framework of IT drug delivery guidelines, then a very rewarding and beneficial therapeutic relationship can be achieved.
References
- Stearns L, Boortz-Marx R, Du Pen S, Friehs G, Gordon M, Halyard M, Herbst L, Kiser J. Intrathecal drug delivery for the management of cancer pain: A multidisciplinary consensus of best clinical practices. J Support Oncol 2005;3:399-408.
- Wallace M, Yaksh TL. Long-term spinal analgesic delivery: A review of the preclinical and clinical literature. Reg Anesth Pain Med 2000;25:117-157.
- Paice JA, Penn RD, Shott S. Intraspinal morphine for chronic pain: a retrospective, multicenter study. J Pain Symptom Manage. 1996;11:71-80.
- Deer T, et al. Intrathecal therapy for cancer and non-malignant pain: patient selection and patient management. Neuromodulation 1999;2:55-66.
- Wallace M, Yakesh TL. Long-term spinal analgesic delivery: a review of the preclinical and clinical literature. Reg Anesth Pain Med 2000;25:117-57.
- Buntin-Muschock C, Phillip L, Moriyama K, Palmer PP. Age-dependent opioid escalation in chronic pain patients. Anesth Analg 2005;100:1740-45.
- Wang Y, Mitchell J, Moriyama K, et al. age-dependent morphine tolerance development in the rat. Anesth Analg 2005;100:1733-39.
- Deer T, et al. Consensus Guidelines for the selection and implantation of patients with noncancer pain for intrathecal drug delivery. Pain Physician 2010;13:E175-E213.
- Deer T, et al. Polyanalgesic consensus conference 2007: recommendations for the management of pain by intrathecal (intraspinal) drug delivery: report of an interdisciplinary expert panel. Neuromodulation 2007;10:300-28.
- Deer T, et al. Polyanalgesic consensus conference 2012: recommendations for the management of pain by intrathecal (intraspinal) drug delivery: report of an interdisciplinary expert panel. Neuromodulation 2012; 15:436-66.
- Deer T, et al. Polyanalgesic consensus conference--2012: consensus on diagnosis, detection, and treatment of catheter-tip granulomas (inflammatory masses). Neuromodulation 2012;15:483-96.
Leave a commentOrder by
Newest on top Oldest on top